Product Images Xyntha
View Photos of Packaging, Labels & Appearance
- Figure - xyntha 01
- Figure - xyntha 02
- Figure - xyntha 03
- Figure - xyntha 04
- Figure - xyntha 05
- Figure - xyntha 06
- Figure - xyntha 07
- Figure - xyntha 08
- Figure - xyntha 09
- Figure - xyntha 10
- Figure - xyntha 11
- Figure - xyntha 12
- Figure - xyntha 13
- Figure - xyntha 14
- Figure - xyntha 15
- Figure - xyntha 16
- Figure - xyntha 17
- Figure - xyntha 18
- Figure - xyntha 19
- Figure - xyntha 20
- Figure - xyntha 21
- Figure - xyntha 22
- Figure - xyntha 23
- Figure - xyntha 24
- Figure - xyntha 25
- Figure - xyntha 26
- Figure - xyntha 27
- Figure - xyntha 28
- Figure - xyntha 29
- Figure - xyntha 30
- Figure - xyntha 31
- Figure - xyntha 32
- Figure - xyntha 33
- Figure - xyntha 34
- Figure - xyntha 35
- Logo - xyntha 36
- PRINCIPAL DISPLAY PANEL - 4ML VIAL LABEL - xyntha 37
- PRINCIPAL DISPLAY PANEL - 250 IU Label - xyntha 38
- PRINCIPAL DISPLAY PANEL - Kit Carton - 250 IU - xyntha 39
- PRINCIPAL DISPLAY PANEL - 500 IU - VIAL LABEL - xyntha 40
- PRINCIPAL DISPLAY PANEL - Kit Carton - 500 IU - xyntha 41
- PRINCIPAL DISPLAY PANEL - 1000 IU - VIAL LABEL - xyntha 42
- PRINCIPAL DISPLAY PANEL - Kit Carton - 1000 IU - xyntha 43
- PRINCIPAL DISPLAY PANEL - 2000 IU - VIAL LABEL - xyntha 44
- PRINCIPAL DISPLAY PANEL - Kit Carton - 2000 IU - xyntha 45
- xyntha 46
Product Label Images
The following 46 images provide visual information about the product associated with Xyntha NDC 58394-015 by Wyeth Biopharma Division Of Wyeth Pharmaceuticals Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 4ML VIAL LABEL - xyntha 37

This is a product made by Wyeth Pharmaceuticals LLC, which is a subsidiary of Pizer Inc located in Philadelphia, PA 19101.*
PRINCIPAL DISPLAY PANEL - 250 IU Label - xyntha 38

This appears to be a description of a disposable syringe used for drug diluent with Xyntha® antihemophilic factor. The syringe contains 0.9% sodium chloride diluent solution and is made by Wyeth Pharmaceuticals LLC. The product is sterile and non-pyrogenic, without any preservatives. However, there isn't any information on the manufacturing date, LOT or EXP numbers.*
PRINCIPAL DISPLAY PANEL - Kit Carton - 250 IU - xyntha 39
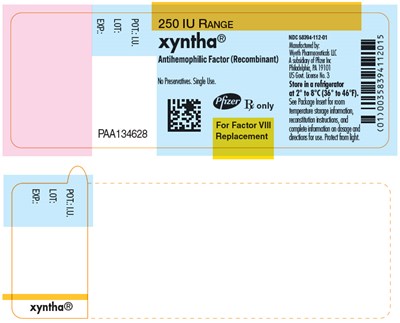
Xyntha® PAA134628 is a medication used for Factor VII replacement in individuals with antihemophilic factor deficiency. It is produced by recombinant technology and has a concentration of 250 IU. The medication is stored in a refrigerator between 2 to 8°C (36° to 46°F). Further dosage information and packaging details are available on the packaging label.*
PRINCIPAL DISPLAY PANEL - Kit Carton - 500 IU - xyntha 41

This is a description of a medication called "xyntha® IR," which is essentially a recombinant antihemophilic factor used to treat patients with hemophilia A. The medication is stored in a refrigerator at a temperature range of 2°C-8°C. The text also contains a reference to "Factor VIll : S PAA134620," which may be a code or identifier related to the medication.*
PRINCIPAL DISPLAY PANEL - Kit Carton - 1000 IU - xyntha 43

This appears to be a description of a medication called Xyntha, which is an antihemophilic factor used as a replacement therapy for Factor VII. The medication should be stored in a refrigerator. The rest of the text is not readable.*
PRINCIPAL DISPLAY PANEL - 2000 IU - VIAL LABEL - xyntha 44

This is a product description for Xyntha, an antihemophilic factor manufactured by Wyeth Pharmaceuticals LLC, a subsidiary of Pfizer Inc. It is used for factor VII replacement therapy and can only be administered intravenously. Each package contains a single-use vial with a prefilled diluent syringe of 1000 IU range, and it is available at room temperature. The text includes some errors and non-English characters.*
PRINCIPAL DISPLAY PANEL - Kit Carton - 2000 IU - xyntha 45

This is a product label for a medication called Antihemophilic Factor (Recombinant) marketed as Xyntha. The medication is used for Factor VIII A treatment and needs to be stored in a refrigerator at a temperature between 2° to 8°C. The National Drug Code for this medication is 58304-115.01.*
xyntha 46

This is a product description of a single-use vial containing 4 mL of antihemophilic factor medicine for intravenous administration. The medicine is manufactured by Wyeth Pharmaceuticals, LLC, which is a subsidiary of Pfizer Inc. The product is for therapy only and is used for factor VII replacement for hemophiliacs. The product has a GTIN number that starts with 003583 and can be stored in room temperature. There is also information regarding storage, handling, usage, and expiration date, which must be checked before use. Some information on the product is unavailable due to errors.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.