Product Images Xyntha
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 51 images provide visual information about the product associated with Xyntha NDC 58394-016 by Wyeth Biopharma Division Of Wyeth Pharmaceuticals Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
xyntha 41

This is a pharmaceutical product manufactured by Wyeth Pharmaceuticals LLC, which is a subsidiary of Pfizer Inc based in Philadelphia, PA 19101.*
xyntha 42

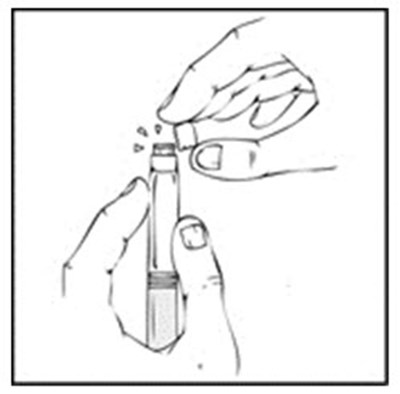
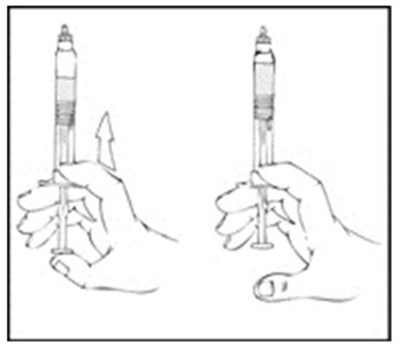
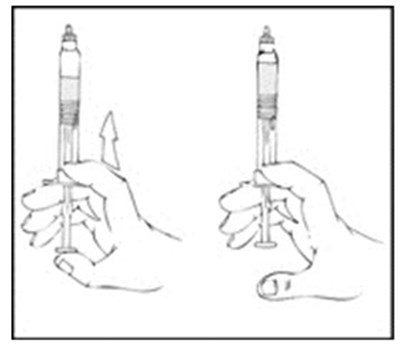
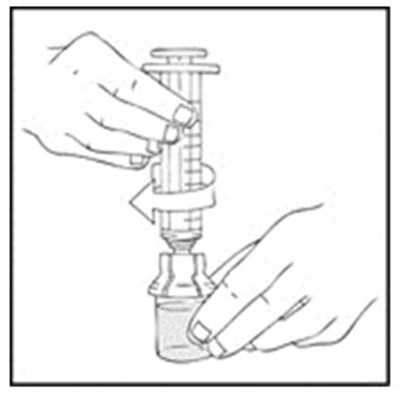
Xyntha Solofuse is a dual-chamber syringe for intravenous administration of recombinant antihemophilic factor. It is preservative-free, for single use only, and should be refrigerated and protected from light. The top chamber contains the antihemophilic factor in a sterile, non-pyrogenic, preservative-free, freeze-dried powder form. The bottom chamber contains 0.8% sodium chloride diluent, which also has no preservatives. Xyntha Solofuse is used for factor Vil replacement. More information can be found in the package insert.*
xyntha 43

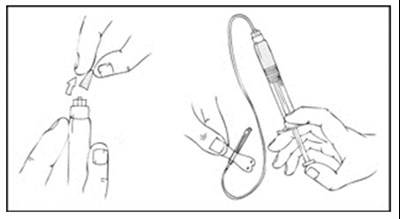
Xyntha is a type of antihemophilic factor used to treat bleeding in patients with hemophilia A. It is available in a single-use, dual-chamber syringe which contains a top chamber and a 0.0% sodium chloride solution-filled diluent chamber. The package includes one plunger, one set of two alcohol swabs, one bandage, one gauze pad, and one package insert. It is manufactured in various locations including Germany, Switzerland, and Belgium. The use of Xyntha is strictly for medical purposes.*
xyntha 44

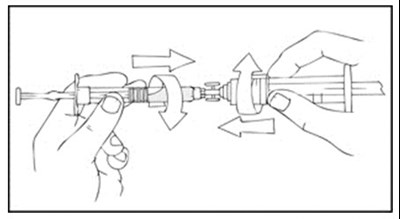
This is a description of a medical product called Xyntha Solofuse, manufactured by Wyeth Pharmaceuticals LLC. It is a dual-chamber syringe containing Antihemophilic Factor used for intravenous administration. The syringe has no preservatives and is for single use only. The top chamber contains sterile, non-pyrogenic, preservative-free, freeze-dried powder while the bottom chamber contains 0.5% Sodium Chloride Dient 4L and no preservatives. The product is refrigerated and should be protected from light. The package insert should be referred for further information.*
xyntha 45

This is a XYNTHA SOLOFUSE kit that includes a prefilled dual-chamber syringe containing XYNTHA and Diluent. It is intended for Factor VIII replacement therapy via intravenous administration. The kit includes sterile XYNTHA top chamber, 4L 0.9% Sodium Chloride in bottom chamber, one extension set, two alcohol wipes, one bandage, one gauze pad, and one sterile cap. The kit is made in several countries and manufactured by Wyeth Pharmaceuticals. The government license number is indicated.*
xyntha 46

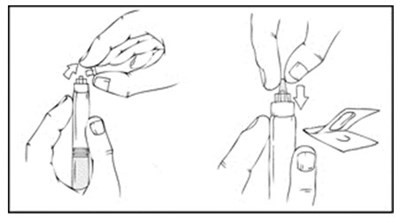
This is a description of Xyntha Solofuse, a medication used for intravenous (IV) administration in patients needing Factor VII replacement. The medication is packaged in a dual-chamber syringe, with the top compartment containing a sterile, nonpyrogenic, preservative-free, lyophilized powder of Antihemophilic Factor (Recombinant). The bottom chamber contains 0.8% Sodium Chioride Dilvent, 4 mL and no preservatives. The medication should be refrigerated and protected from light. More information about the medication can be found in the package insert. The text also provides the PAA number and NDC code of the medication, as well as the manufacturer's name and address.*
xyntha 47

The text seems to be a product description for a XYNTHA SOLOFUSE kit. The kit contains a prefilled syringe with XYNTHA and diluent, a plunger for assembly, a serial infusion set, two alcohol wipes, a bandage, an EZ package insert, and a converted cap. It is manufactured in Germany and Switzerland. The text also includes some identification numbers and symbols.*
xyntha 48

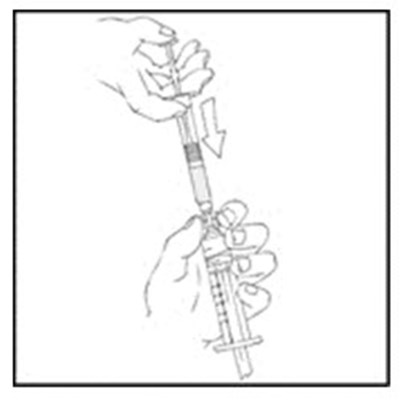
This is a description of Xyntha Solofuse, a medication used for intravenous administration, that contains the Aniihemophilc Factor (Recombinant) in a sterile, non-pyrogenic, preservative-free, freeze-dried powder form in the top chamber of a dual-chamber syringe. The bottom chamber of the syringe contains a diluent with 0.9% sodium chloride, devoid of preservatives. This syringe is for single-use, and requires refrigeration while being protected from light. This medication is designed for Factor VII replacement. More detailed information is available in the package insert.*
xyntha 49

This is a description of a medical product called XYNTHA SOLOFUSE kit. It contains a single-use, prefilled dual-chamber syringe with XYNTHA antihemophilic factor (recombinant) and diluent for intravenous administration. It has a plunge rod made in Switzerland and manufactured by Wyeth Pharmaceuticals. The kit also contains other accessories like a gas pad, assembly, and four L 0.9% Sodium Chloride bottom chamber. Some of the text is not readable due to errors.*
xyntha 50

This is a description of Xyntha® Solofuse®, a dual-chamber syringe containing a sterile, nonpyrogenic, preservative-free, freeze-dried powder of Antihemophilic Factor (Recombinant) in the top chamber and a 0.9% Sodium Chloride Diluent in the bottom chamber that contains no preservatives. The syringe is used for IV administration and is a single-use product that should be refrigerated and protected from light. The package insert should be consulted for further information. The text also includes product information such as Lot, Exp., Pot., NDC code, and the manufacturer's name and address. The product is meant for Factor Vll Replacement.*
xyntha 51

This is a kit of XYNTHA SOLOFUSE which includes a single-use, dual-chamber syringe prefilled with XYNTHA antihemophilic factor (recombinant) and diluent for intravenous administration. The syringe contains 3000 IU range and comes with a single-vopald syringe containing 4 ol of 09% Sodium Chioridinbotim, one lnge o o assembly, one tartenfsion e, one dlcchol wsbs, one bandage, one gauzspad, one vertd serls cap, and rclone in the package nsart. The manufacturer of the product is WysthPhamacauticals LLC, a subsidiary of Pz Inc Plunge Hod, and it is made in Germany, Switzerland, and Belgium.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.