Product Images Losartan Potatssium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Losartan Potatssium NDC 58517-240 by New Horizon Rx Group, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
3457501e-figure-02 - 3457501e figure 02

This appears to be a result summary of a clinical trial comparing the medications Atenolol and Losartan Potassium. The data shows that 9% of the patients achieved the primary endpoint, and the adjusted risk reduction was 13% with a p-value of 0.021. The table shows various study months and potentially some numerical results associated with them, but without more context it is difficult to determine their meaning.*
3457501e-figure-03 - 3457501e figure 03

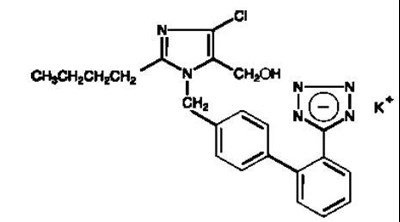
This text is not-available since it only lists medication names and some symbols.*
3457501e-figure-05 - 3457501e figure 05
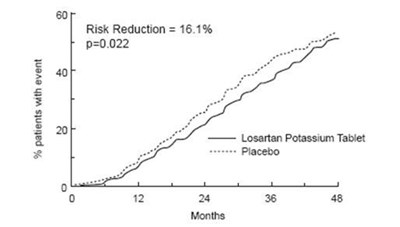
This document is describing the results of a clinical trial involving Losartan Potassium Tablet and a placebo. It states that 9% of patients experienced an event, and that there was a 16.1% risk reduction with Losartan Potassium Tablet compared to the placebo. The value of p is 0.022. The meaning of "12 2 % 48" is not clear from this text and may be a scanning error.*
3457501e-figure-06 - 3457501e figure 06

This is a description of Losartan Potassium Tablets, USP with NDC number 58517-240-30, containing 30 tablets of 0mg strength. The remaining text seems to be garbled or not relevant.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.