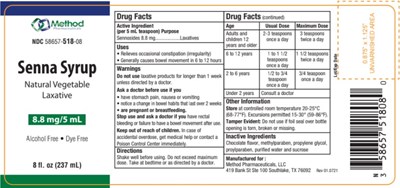
FDA Label for Senna Syrup
View Indications, Usage & Precautions
Senna Syrup Product Label
The following document was submitted to the FDA by the labeler of this product Method Pharmaceuticals. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredients
Sennosides 8.8 mg
Purpose
Laxative
Warning
Do not use laxative products for longer than 1 week
unless directed by a doctor.
Uses
• Relieves occasional constipation (irregularity)
• Generally causes bowel movement in 6 to 12 hours
Ask A Doctor Before Use If You
• have stomach pain, nausea or vomiting
• notice a change in bowel habits that last over 2 weeks
• are
pregnant or breastfeeding.
Stop Use And Ask A Doctor
if you have rectal bleeding or failure to have a bowel movement after use.
Keep Out Of Reach Of Children
In case of accidental overdose, get medical help or contact a Poison Control Center immediately.
Directions
Shake well before using. Do not exceed maximum dose. Take at bedtime or as directed by a doctor.
Other Safety Information
Store at controlled room temperature 20-25°C(68-77°F). Excursions permitted 15-30° (59-86°F).
Tamper Evident: Do not use if foil seal over bottle opening is torn, broken or missing.
Inactive Ingredients
Chocolate flavor, methylparaben, propylene glycol, proylparaben, purified water and sucrose
Dosage
| Age | Usual Dose | Maximum Dose |
| Adults and Children 12 years and older | 2-3 teaspoons once a day | 3 teaspoons twice a day |
| 6-12 years | 1 to 1 1/2 teaspoons once a day | 1 1/2 teaspoons twice a day |
| 2-6 years | 1/2 to 3/4 teaspoon once a day | 3/4 teaspoons twice a day |
| under 2 years | Consult a doctor |
Senna Syrup
Manufactured for :
Method Pharmaceuticals, LLC
419 Bank St Ste 100 Southlake, TX 76092
Rev 01.0721
* Please review the disclaimer below.