Product Images Abilify Maintena
View Photos of Packaging, Labels & Appearance
- PRINCIPAL DISPLAY PANEL - 300 mg Syringe Carton - abilify 300mg syringe carton
- PRINCIPAL DISPLAY PANEL - 300 mg Syringe Label - abilify 300mg syringe label
- PRINCIPAL DISPLAY PANEL - 300 mg Kit Carton - abilify 300mg vial carton
- PRINCIPAL DISPLAY PANEL - 300 mg Vial Label - abilify 300mg vial label
- abilify 400mg syringe carton
- PRINCIPAL DISPLAY PANEL - 400 mg Syringe Label - abilify 400mg syringe label
- PRINCIPAL DISPLAY PANEL - 400 mg Kit Carton - abilify 400mg vial carton
- PRINCIPAL DISPLAY PANEL - 400 mg Vial Label - abilify 400mg vial label
- PRINCIPAL DISPLAY PANEL - 5 mL Vial Label - abilify 5ml water label
- Chemical Structure - abilify chem structure
- Figure 1 - abilify figure 01
- Figure 2 - abilify figure 02
- Figure 3 - abilify figure 03
- Figure 4 - abilify figure 04
- Figure 5 - abilify figure 05
- Figure 6 - abilify figure 06
- Figure 7 - abilify figure 07
- Figure 8 - abilify figure 08
- Figure 9 - abilify figure 09
- Figure 10 - abilify figure 10
- Figure 11 - abilify figure 11
- Figure 12 - abilify figure 12
- Figure 13 - abilify figure 13
- Figure 14 - abilify figure 14
- Figure 15 - abilify figure 15
- Figure 16 - abilify figure 16
- Figure 17 - abilify figure 17
- Figure 18 - abilify figure 18
- Figure 19 - abilify figure 19
- Figure 20 - abilify figure 20
- Figure 21 - abilify figure 21
- Figure 22 - abilify figure 22
- Figure 23 - abilify figure 23
- Figure 24 - abilify figure 24
- Figure 25 - abilify figure 25
- Figure 26 - abilify figure 26
Product Label Images
The following 36 images provide visual information about the product associated with Abilify Maintena NDC 59148-019 by Otsuka America Pharmaceutical, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
abilify 400mg syringe carton

Abilify Maintena is a medication that can be administered through deltoid or gluteal intramuscular injection only using a dual-chamber syringe. Each injection must be administered by a healthcare professional and an enclosed Medication Guide must be dispensed to each patient. The NDC number for this product is 5914807280. Other text in the output is not legible and cannot be used to provide any additional useful information.*
PRINCIPAL DISPLAY PANEL - 400 mg Kit Carton - abilify 400mg vial carton

Abilify Maintena is an extended-release injectable suspension of eripiprazol. The medication is used for intramuscular injection only into the deltoid or gluteal muscles. Each vial is for single use only and must be discarded if any portion remains unused. The drug requires reconstitution by adding 1.9 mL of sterile water, and excess water is left in the vial. Abilify Maintena must only be administered by a healthcare professional. Keep it away from children.*
PRINCIPAL DISPLAY PANEL - 400 mg Vial Label - abilify 400mg vial label

Abilify Maintena is a medication used for intramuscular injection only. It is a single-use sterile medication, and any unused portion should be discarded. The dosage should be determined by consulting the package insert. No other useful information is available due to the poor quality.*
PRINCIPAL DISPLAY PANEL - 5 mL Vial Label - abilify 5ml water label

This is a sterile water for injection, which comes in 5mL and is meant to be a single-use product; any unused portion must be discarded. It should be stored at a temperature between 15 °C and 30 °C, as long as the temperature does not exceed 25 °C. It is only available as a prescription product and is distributed by Lundbeck.*
Figure 1 - abilify figure 01

After plunger rod is at complete stop, "middle stopper will be at the indicator line.*
Figure 2 - abilify figure 02

This is a description of a substance that is uniform and milky white in color. It has a suggested use time of 20 seconds after reconstitution and should be used within 30 minutes of reconstitution.*
Figure 4 - abilify figure 04

This text provides information on recommended injection site needle sizes based on body type for the deltoid and gluteus muscles. The size varies depending on body type, with a recommended 1 inch (23a) needle for non-obese individuals in the deltoid muscle and a 1.5 inch (22q) needle for non-obese individuals in the gluteus muscle. For obese individuals, a 1.5 inch (22q) needle is recommended for the deltoid muscle and a 2 inch (216) needle is recommended for the gluteus muscle.*
Figure 7 - abilify figure 07

This text appears to be instructions on how to expel air from a needle base. It advises expelling air until the suspension fills the needle base, and if there is difficulty, to check that the plunger rod is rotated to a complete stop.*
Figure 19 - abilify figure 19
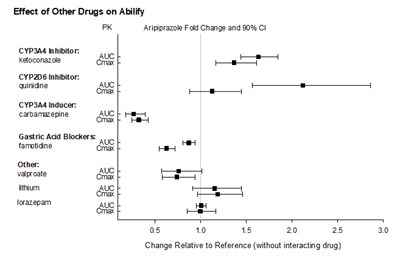
The text appears to be related to the effect of other drugs on Abilify. Specifically, the text mentions the influence of ketoconazole, CaP3A4 inducers, Cavrazagne AU, and gastric acid blockers on the Cmax levels of Aripiprazole, along with fold change and 90% CI values. The rest of the text seems to be incomplete and not related to the main topic.*
Figure 20 - abilify figure 20
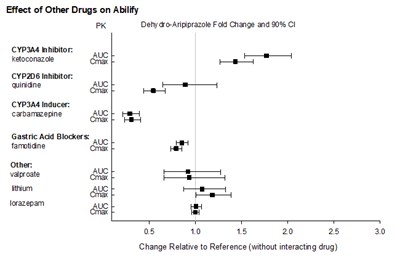
This text provides information on the effect of other drugs on Abilify by showing the fold change in PK Dehy aro-Aripiprazole and 80% CI caused by different types of inhibitors and inducers such as CYP3A4, CYP2D6, gastric acid blockers, and others. The text also includes the relative change to the reference without the interacting drug.*
Figure 21 - abilify figure 21
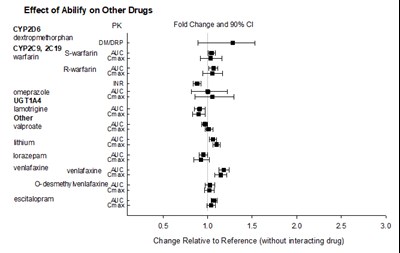
This text provides information about the effect of Abilify on other drugs. It includes a list of different drugs such as warfarin, omeprazole, valproate, lithium, lorazepam, and escitalopram, among others. The text presents a chart showing the fold change and 90% confidence interval for each drug. The chart also displays changes relative to the reference without interacting drug.*
Figure 22 - abilify figure 22

This appears to be a table of factors related to drug metabolism and dosing in different populations, including differences in metabolism based on genetics (CYPANS, poor vs extensive metabolizers), gender, age, and degree of liver and kidney impairment. The table also appears to include information on a specific drug called Aripiprazole and how it is affected by these factors. The numbers in the table seem to represent fold changes and confidence intervals.*
Figure 24 - abilify figure 24
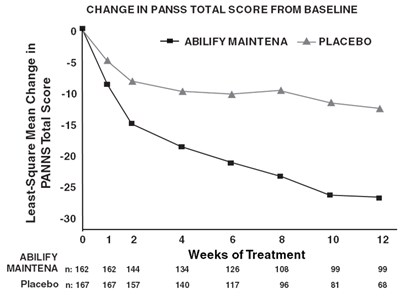
The text describes a chart showing the change in PANSS total score from baseline over time for two treatments: Abilify Maintena and placebo. The chart shows a baseline score of 0 and scores at weeks 2, 4, 6, 8, 10, and 12. The Abilify Maintena group had 162 participants and the placebo group had 167. The chart shows that the Abilify Maintena group had a decrease in total score and the placebo group had a smaller decrease in total score.*
Figure 25 - abilify figure 25

This text contains bar graphs and tables showing the results of a study comparing Abilify Maintena and placebo in preventing relapse of schizophrenia. The graphs show the percentage of subjects who did not relapse over time, and the tables show the number of subjects still at risk at each time point.*
Figure 26 - abilify figure 26

The text describes a graph showing the proportion of subjects with recurrence over time. The graph shows percentages ranging from 0% to 80%, with corresponding numbers of subjects at risk listed below. The time to recurrence is also measured and compared between two groups: Abity Maintena and Placebo. There is not enough information given to provide further details.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




















