FDA Label for Allergy And Sinus Relief
View Indications, Usage & Precautions
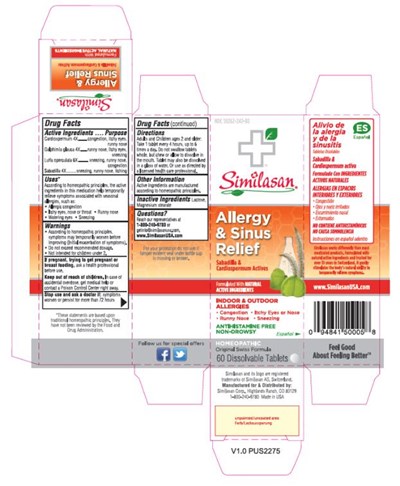
Allergy And Sinus Relief Product Label
The following document was submitted to the FDA by the labeler of this product Similasan Corporation. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredients
Cardiospermum 4X
Galphimia glauca 4X
Luffa operculata 6X
Sabadilla 4X
Purpose
congestion, itchy eyes, runny nose
runny nose, itchy eyes, sneezing
sneezing, runny nose, congestion
sneezing, runny nose, itching
Uses*
According to homeopathic principles, the active ingredients in this medication help temporarily relieve symptoms associated with seasonal allergies, such as:
• Allergic congestion
• Itchy eyes, nose or throat
• Runny nose
• Watering eyes
• Sneezing
Warnings
• According to homeopathic principles, symptoms may temporarily worsen before improving (Initial exacerbation of symptoms).
• Do not exceed recommended dosage.
• Not intended for children under 2.
Keep Out Of Reach Of Children.
In case of accidental overdose, get medical help or contact a Poison Control Center right away.
Stop Use And Ask A Doctor If:
symptoms worsen or persist for more than 72 hours
Directions
Adults and Children ages 2 and older: Take 1 tablet every 4 hours, up to 6 times a day. Do not swallow tablets whole, but chew or allow to dissolve in the mouth. Tablet may also be dissolved in a glass of water. Or use as directed by a licensed health care professional.
Other Information
Active ingredients are manufactured according to homeopathic principles.
Inactive Ingredients
Lactose, Magnesium stearate
Questions?
Reach our representatives at 1-800-240-9780 or [email protected]. www.SimilasanUSA.com
*These statements are based upon traditional homeopathic principles. They have not been reviewed by the Food and Drug Administration.
Principal Display Panel
NDC 59262-242-30
Similasan
Allergy
& Sinus
Relief
60 Dissolvable Tablets
* Please review the disclaimer below.