Product Images Retacrit
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 31 images provide visual information about the product associated with Retacrit NDC 59353-004 by Vifor (international) Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - retacrit 01
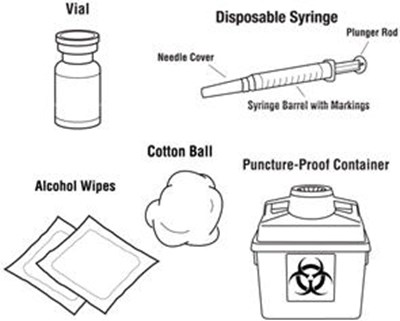
This text is not enough to generate a useful description. It only contains two words that can be found on medical equipment: 'Vial' and 'Disposable Syringe'. Without context, it's not possible to give a meaningful description.*
PRINCIPAL DISPLAY PANEL - 2,000 Units/mL Vial Label - retacrit 17

This appears to be a label for a medication. The medication is Epoetin Alfa-epbx, a recombinant produced by Pfizer Company, and distributed by Vifor Pharma in a single-dose vial containing 1 mL solution with a strength of 2,111,111 Units/mL. The dosage instructions cannot be determined from the available text. The medication is US licensed with the number 1974. The label seems to have a barcode.*
PRINCIPAL DISPLAY PANEL - 2,000 Units/mL Vial Carton - retacrit 18
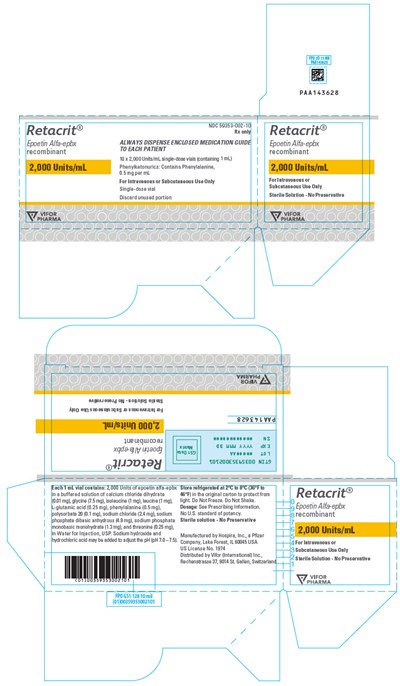
This is a medicine called Retacrit which is meant for intravenous or subcutaneous use only. It is a recombinant Epoetin that is available in single-dose vials. The medication guide enclosed with the vial must be followed for proper use. The solution is not meant to be preserved and any unused portion should be discarded properly. The dosage and other information are mentioned in the prescribing information. The medicine is manufactured by Hospis, Inc and distributed by Vior (International) in Switzerland.*
PRINCIPAL DISPLAY PANEL - 3,000 Units/mL Vial Label - retacrit 19

This appears to be a medication label for Epoetin Alfa-epbx, a recombinant drug produced by Pfizer and distributed by Vifor Pharma. The label includes information on dosage and prescribing, as well as the drug concentration and packaging.*
PRINCIPAL DISPLAY PANEL - 3,000 Units/mL Vial Carton - retacrit 20
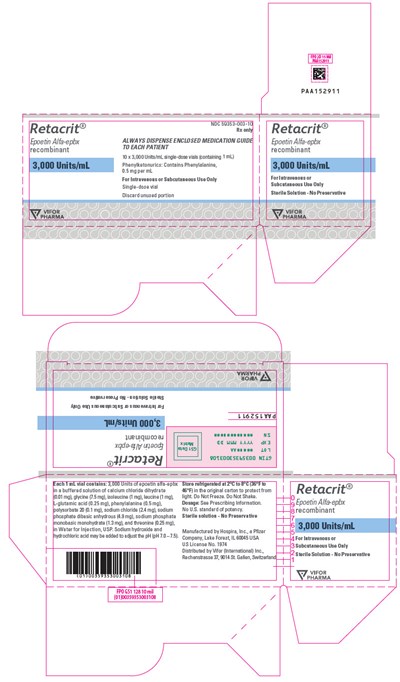
Retacrit® is a medicine that comes in a single-dose vial and is recommended for intravenous use only. The medicine should be discarded after use. The vial contains recombinant Epoetin Alfa and is dispensed to each patient. The vial has a potency of 33 Units/mL and contains Phenyketonurics, which is a substance that cannot be taken by certain individuals. The doses of the medicine may vary and should be taken as per the doctor's prescription. The vial has polysorbate 20, sodium chloride, and phosphate dibasic. There is also a sterile solution that is formulated without any preservatives. The medicine is manufactured by a company located in USA.*
PRINCIPAL DISPLAY PANEL - 4,000 Units/mL Vial Label - retacrit 21

This appears to be a label for a medication called Epoetin Alfa-epbx, manufactured by Hospira, Inc., a subsidiary of Pfizer. However, the text is not complete and some characters are not readable, which makes it difficult to provide a more specific description.*
PRINCIPAL DISPLAY PANEL - 4,000 Units/mL Vial Carton - retacrit 22

This is a product label for various medications including Retacrit and Epostin Alfepbx. These medications are for intravenous or subcutaneous use only, and come in single-dose vials. The label provides instructions for dosage and administration. The company is based in Switzerland and the medications are distributed by Vifor International.*
PRINCIPAL DISPLAY PANEL - 10,000 Units/mL Vial Carton - retacrit 24
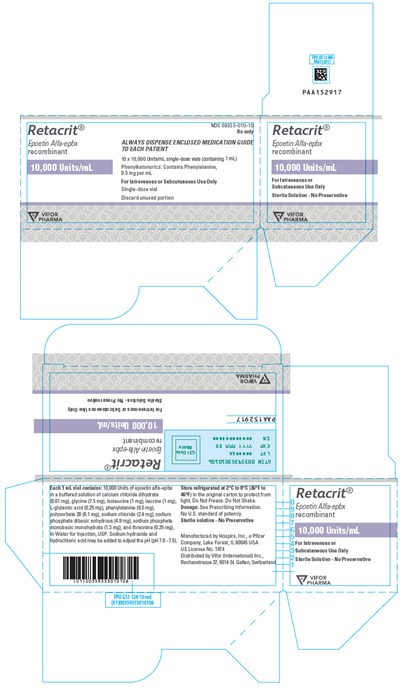
Retacrit is a recombinant Epoetin Alfa-epbx in a 10,000 Units/mL concentration. It comes in a single-dose vial, intended for intravenous or subcutaneous use only. Phanykatonurics should be cautious as it contains Phanylalaning, 05mg per mL. Patients should be provided with an enclosed medication guide. The medication is manufactured by Hospira, a Pfizer Company in Lake Forest, L6004 USA, and distributed by Vviar International inc. in its original carton to protect it. The dosage and other information should be as per the prescribing information. The vial should be discarded after use, and any unused portion should be discarded.*
retacrit 27

This is a label for a medication manufactured by Hospira, which is a Pfizer company, based in Lake Forest, USA. The dosage information is not provided and needs to be obtained from the prescribing information. The medication is distributed by Vifor (International) Inc., located in St Gallen, Switzerland. PAA158676 is mentioned, but its context is not clear. The label also contains a barcode.*
retacrit 28
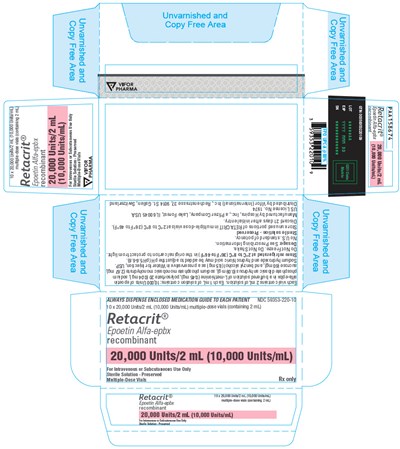
Retacrit® (Epoetin Alfa-epbx recombinant) is a sterile solution provided in 20,000 Units/2 mL (10,000 Units/mL) multiple-dose vials for intravenous or subcutaneous use. The vials have a copy-free area and come with an enclosed medication guide to be dispensed to each patient. The text does not provide additional information.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




















