Product Images Ryaltris
View Photos of Packaging, Labels & Appearance
- CTN.jpg - CTN
- FigureA.jpg - FigureA
- FigureB.jpg - FigureB
- FigureC.jpg - FigureC
- FigureD.jpg - FigureD
- FigureE.jpg - FigureE
- FigureF.jpg - FigureF
- FigureG.jpg - FigureG
- FigureH.jpg - FigureH
- FigureI.jpg - FigureI
- FigureJ.jpg - FigureJ
- FigureK.jpg - FigureK
- FigureL.jpg - FigureL
- LBL.jpg - LBL
- Structure-Mometasone.jpg - Structure Mometasone
- Structure-Olopatadine.jpg - Structure Olopatadine
Product Label Images
The following 16 images provide visual information about the product associated with Ryaltris NDC 59467-700 by Hikma Specialty Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
CTN.jpg - CTN
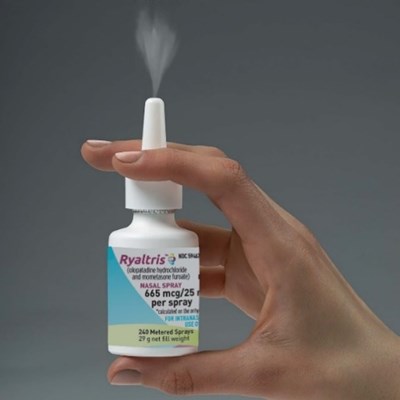
Ryaltris is a nasal spray for intranasal use only. It contains olopatadine hydrochloride and mometasone furoate at 665 mcg/25 mcg per spray. Each metered spray delivers 100 mg of suspension containing 865 mcg olopatadine hydrochloride, equivalent to 600 mcg olopatadine as base, and 25 mcg mometasone furoate. The contents are an aqueous suspension containing various ingredients. It is recommended to shake the bottle well before each use. The initial priming requires spraying until a fine mist appears, and the reprime requires spraying until a fine mist appears only if Ryaltris has not been used for 14 days. The dosage should be prescribed by a doctor. The storage instructions advise not to store in the freezer or refrigerator, and keep it out of reach of children. The dosing instructions include blowing one's nose and tilting the head while spraying while inhaling gently. Two sprays per nostril are recommended, and Ryaltris should be discarded after 240 actuations. The package contains 29g net fill weight and 240 metered sprays. For additional assistance, one can call 1-800-962-8364 or visit WRYALTRIS.com. The product is distributed by Hikma Specialty USA Inc, Columbus, OH 63228.*
FigureA.jpg - FigureA
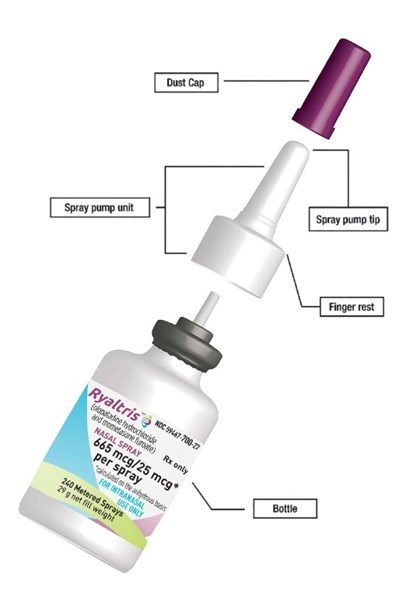
This is likely a list of items related to a "spray pump unit". It includes a "dust cap", "spray pump tip", "finger rest", and a "bottle". The context and purpose of these items cannot be determined without further information.*
LBL.jpg - LBL

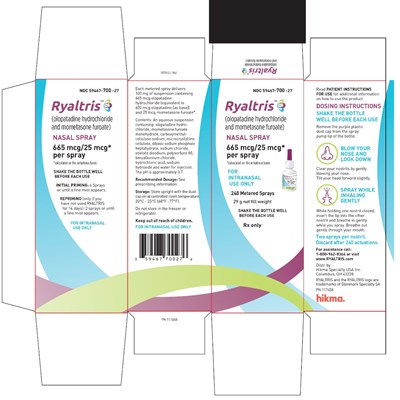
This text provides instructions and information on using RYALTRIS nasal spray for intranasal use only. The user is instructed to prime the spray with six sprays or until a fine mist appears before each use and to use two sprays or until a fine mist appears following a storage period of more than 14 days. The recommended dosage for the prescription medication is not provided and users are instructed to refer to the prescribing information. Storage instructions indicate that the nasal spray should be kept upright with the dust cap on at a controlled room temperature and should not be stored in the refrigerator or freezer. The product contains olopatadine hydrochloride and mometasone furoate and features 665 mcg/25 mcg per spray with a net fill weight of 29 g.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.