FDA Label for Tena
View Indications, Usage & Precautions
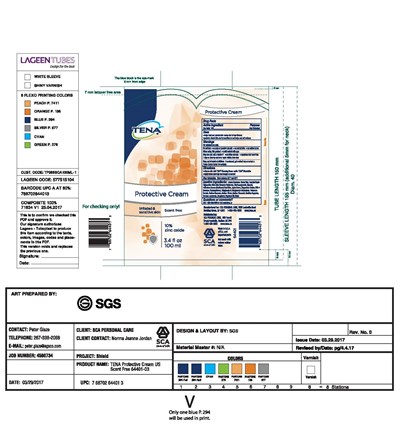
Tena Product Label
The following document was submitted to the FDA by the labeler of this product Sca Personal Care Inc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Otc - Active Ingredient
Active Ingredient
Zinc Oxide 10%
Otc - Purpose
Purpose
Skin Protectant
Indications & Usage
Uses
•helps treat and prevent skin rashes due to incontinence
•protects chafed skin due to incontinence and helps seal out wetness
Warnings
Warnings
For external use only.
Do not use •on deep or puncture wounds •on animal bites •on serious burns
When using this product •avoid contact with eyes
Stop use and ask a doctor if •condition worsens •symptoms last more than
7 days or clear up and occur again within a few days
Otc - Keep Out Of Reach Of Children
Keep out of reach of children. If swallowed, get medical help or contact a
Poison Control Center immediately
Dosage & Administration
Directions
•cleanse skin with TENA® Cleansing Cream and/or TENA® Washcloths
•apply cream sparingly and reapply as needed
Storage And Handling
Other information Store between 32° F and 85° F
Inactive Ingredient
Inactive ingredients: Acacia Decurrens Flower Wax, Caprylic/Capric Triglyceride, Citric Acid, Dicaprylyl Carbonate,
Ethylhexylglycerin, Glycerin, Helianthus Annuus (Sunflower) Seed Wax, Jojoba Esters, Magnesium Sulfate, PEG-8 Beeswax, Polyglycerin-3,
Polyglyceryl-3 Ricinoleate, Potassium Sorbate, Sodium Benzoate, Sodium Citrate, Sodium Gluconate, Sorbitan Caprylate, Sorbitan
Isostearate, Tocopherol, Vegetable Oil, Water.
Package Label.Principal Display Panel
* Please review the disclaimer below.