Product Images Imatinib Mesylate
View Photos of Packaging, Labels & Appearance
- Figure 1: Progression Free Survival (ITT Principle) - imatinib fig1
- Figure 2: Time to Progression to AP or BC (ITT Principle) - imatinib fig2
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg - (90 Tablets Bottle) - imatinib fig3
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 400 mg - (30 Tablets Bottle) - imatinib fig4
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 400 mg (Blister Carton Label) - imatinib fig5
- imatinib fig6
- imatinib fig7
- imatinib fig8
- Chemical Structure - imatinib str
Product Label Images
The following 9 images provide visual information about the product associated with Imatinib Mesylate NDC 59651-241 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1: Progression Free Survival (ITT Principle) - imatinib fig1
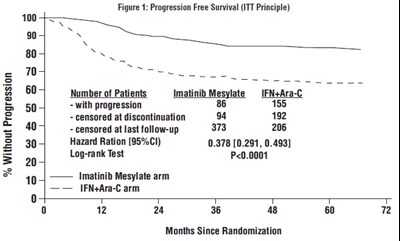
The text describes a figure (Figure 1) showing the Progression-Free Survival of patients with Imatinib Mesylate arm and IFN+Ara-C arm. The number of patients with progression, censored at discontinuation, and censored at last follow-up are also shown. The Hazard Ratio and Log-rank Test statistic are also provided. The x-axis shows the number of months since randomization.*
Figure 2: Time to Progression to AP or BC (ITT Principle) - imatinib fig2

This is a figure that shows the time taken for patients to progress to AP or BC using the ITT principle. The data is presented in a graph with the number of patients who progressed and were censored at discontinuation or last follow-up, respectively. The hazard ratio and p-value are also provided. The graph shows that the Imatinib Mesylate arm had a lower risk of progression compared to the IFN+Ara-C arm. The x-axis represents the time in months since randomization, while the y-axis represents the percentage of patients.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 400 mg (Blister Carton Label) - imatinib fig5
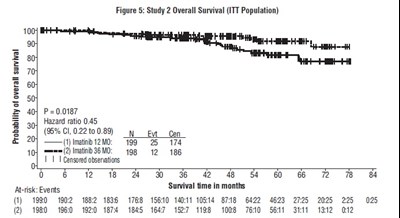
The text describes Figure 5 which shows the overall survival for a study 2 with ITT population. The X-axis shows the time in months and the Y-axis shows the percentage of survival. The data shows the number of patients observed under censorship and the one that has been assigned the treatment. The data also provides the hazard ratio of 0.45 and p-value of 0.0187.*
imatinib fig6

Imatinib Mesylate Tablets by Aurobindo Pharma USA, Inc. is a prescription medicine containing 90 film-coated tablets. Each tablet contains Imatinib mesylate equivalent to 100mg of imatinib free base. It is used as per the instructions given in the package insert. It should be stored between 20°C to 25°C and protect it from moisture. It should be dispensed in a light container, USP. The package is distributed by Aurobindo Pharma USA, Inc. and made in India.*
imatinib fig8

This is a package of Imatinib Mesylate Tablets, with each tablet containing 400 mg of the active ingredient. The package contains 30 unit-dose tablets and should be stored at room temperature between 20-25°C. The tablets are distributed by Aurobindo Pharma USA, Inc. in East Windsor, NJ and are made in India. The package is only available with a prescription, and it is important to keep it out of the reach of children.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.



