Product Images Timolol Maleate
View Photos of Packaging, Labels & Appearance
- Figure 1 - timolo fig1
- International Study - timolo fig2
- Figure A - timolo fig3
- Figure B - timolo fig4
- Figure C - timolo fig5
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 0.5% Container Label - timolo fig6
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 0.5% – Container-Carton Label - timolo fig7
- chemical structure - timolo str
Product Label Images
The following 8 images provide visual information about the product associated with Timolol Maleate NDC 59651-313 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - timolo fig1
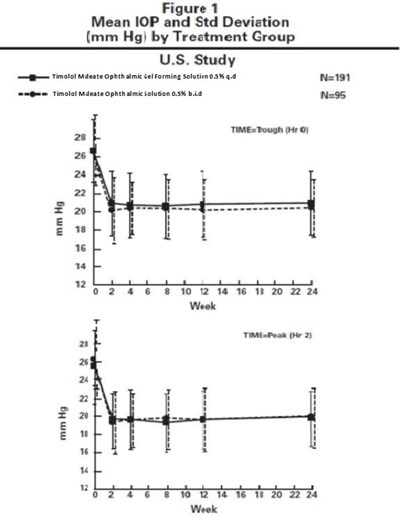
This is a graph showing the Mean Intraocular Pressure (IOP) and standard deviation in millimeters of mercury (mmHg) for two treatment groups in a US study with 191 participants. The treatment groups received either Timolol Maleate Ophthalmic Solution 0.5% once daily or Timolol Maleate Ophthalmic Solution 0.5% twice daily. The x-axis shows the week of the study, while the y-axis shows the mmHg values.*
International Study - timolo fig2

This appears to be a chart or a table with numerical values related to an International Study involving 226 participants and a drug called Timolol Maleate. The drug is administered in two forms: Ophthalmic Gel-Forming Solution (.4) and Ophthalmic Olufon (05%) twice a day. There is data on blood pressure (mm Hg) at Trough (Hr 0) and Peak (Hr 2) for the participants over the course of several weeks. The chart appears to be divided into subgroups labeled R, S, I, NS, IS, FS, N, RN, and Week.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 0.5% Container Label - timolo fig6

This is a description for a medication called OBINDO used for ophthalmic purposes (in the eye) once a day. The package insert should also be consulted. The formulation contains Timolol Maleate in a gel-forming solution and should be stored between 15°C to 25°C. The NDC code is 59651-313-05. The medication is produced by Pyrohindo Pharma USA, Inc. The text suggests that the medication is available in India and is for topical use only.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 0.5% – Container-Carton Label - timolo fig7

This is a prescription medication with the NDC 59651-313-05, which is a solution containing Timolol Maleate at 0.5% concentration ( 6.8 mg/mL). It is topically applied in the eye and is sterile. The solution contains preservatives and inactive ingredients, including mannitol and water, while the preservative is benzododecinum bromide. The recommended dosage as per instructions is 1 drop in the eye once a day. The product needs proper storage to prevent freezing and contamination. It is distributed by Aurobindo Pharma USA and is made in India. There is no additional information available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.



