Product Images Dasatinib
View Photos of Packaging, Labels & Appearance
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 mg (60 Tablet Bottle) - dasatinib fig1
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg 15 (1x15) Unit-dose Tablets - dasatinib fig10
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 140 mg (30 Tablet Bottle) - dasatinib fig11
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 140 mg 15 (1x15) Unit-dose Tablets - dasatinib fig12
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 mg 15 (1x15) Unit-dose Tablets - dasatinib fig2
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg (60 Tablet Bottle) - dasatinib fig3
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg 15 (1x15) Unit-dose Tablets - dasatinib fig4
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 70 mg (60 Tablet Bottle) - dasatinib fig5
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 70 mg 15 (1x15) Unit-dose Tablets - dasatinib fig6
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 80 mg (30 Tablet Bottle) - dasatinib fig7
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 80 mg 15 (1x15) Unit-dose Tablets - dasatinib fig8
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg (30 Tablet Bottle) - dasatinib fig9
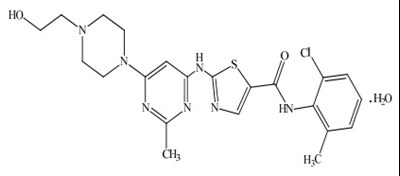
- chemical-structure - dasatinib str
Product Label Images
The following 13 images provide visual information about the product associated with Dasatinib NDC 59651-546 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 mg (60 Tablet Bottle) - dasatinib fig1

This is a description of Dasatinib Tablets, each containing 20 mg of Dasatinib, issued by Aurobindo Pharma USA, Inc. It is advised not to crush, cut, or chew the tablets but to swallow them whole. The package insert contains dosage instructions, directions for use, and precautions. The tablets should be stored at 20° to 25°C and kept away from temperatures of 15° to 30°C. Do not use if the inner seal of the bottle is broken or missing. This medication is distributed by Aurobindo Pharma USA, Inc. from East Windsor, NJ, and manufactured in India.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg 15 (1x15) Unit-dose Tablets - dasatinib fig10

This text provides information regarding Dasatinib Tablets, with each film-coated tablet containing 100 mg of the medication. The usual dosage instructions, storage conditions, and precautions are highlighted. It states not to crush, cut, or chew the tablet but to swallow it whole. The package is not child-resistant. Rxonly is mentioned on the package, and storage should be done at 20°to 25°C (68°to 77°F).*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 140 mg 15 (1x15) Unit-dose Tablets - dasatinib fig12

Each film-coated tablet contains 140 mg of Dasatinib. The usual dosage should be followed as per the instructions provided in the package insert. Store the tablets in a temperature range of 20° to 25° (68° to 77°F), with excursions permitted between 59° to 86°F. These Dasatinib tablets come in a pack of 15 (1x15) unit-dose tablets. Do not crush, cut, or chew the tablets; swallow them whole. Remember, this product is available by prescription only.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 mg 15 (1x15) Unit-dose Tablets - dasatinib fig2

This is a description of Dasatinib Tablets, each pack containing 15 tablets in a unit dose pack. The tablets should not be crushed, cut, or chewed; they should be swallowed whole. The recommended dosage is specified on the package, and the storage temperature range is provided. The medication must be used based on the prescription and instructions from the healthcare provider.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg (60 Tablet Bottle) - dasatinib fig3

This is an evaluation text.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg 15 (1x15) Unit-dose Tablets - dasatinib fig4

This text provides information about Dasatinib tablets containing 50 mg of the active ingredient. It includes guidelines on dosage, storage conditions, and usage instructions. The tablets should be swallowed whole and not crushed, cut, or chewed. The product is offered in a unit-dose package and is prescription-only. Storage should be at 20° to 25°C with permissible excursions between 15° to 30°C. Additional details such as package insert, dosing instructions, and precautions are available.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 70 mg (60 Tablet Bottle) - dasatinib fig5

Description: This text provides information about 70 mg Dasatinib film-coated tablets distributed by Aurobindo Pharma USA, Inc. It includes details on dosage, storage instructions, precautions, and not to be used if inner seal is broken. The text also mentions the product's NDC number, dosage instructions, and company address in East Windsor, NJ.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 70 mg 15 (1x15) Unit-dose Tablets - dasatinib fig6

The provided text seems to be labeling information for Dasatinib 70 mg film-coated tablets. It mentions the usual dosage, storage conditions, and instructions not to crush, cut, or chew the tablets but swallow them whole. The package contains 15 unit-dose tablets and is not child-resistant. It also includes the National Drug Code (NDC) for identification. For detailed dosing information and precautions, refer to the package insert.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 80 mg 15 (1x15) Unit-dose Tablets - dasatinib fig8

This is a product description for Dasatinib Tablets, containing 80 mg of Dasatinib per film-coated tablet. The usual dosage instructions are included in the package insert. It is advised to store the tablets at 20° to 25°C (68° to 77°F) with excursions permitted between 15° to 30°C (59° to 86°F). Each pack contains 15 unit-dose tablets, and it is essential not to crush, cut, or chew the tablets; they should be swallowed whole.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg (30 Tablet Bottle) - dasatinib fig9

This text provides information about a medication called "Dasatinib Tablets" containing 100 mg of Cininin distributed by Aurobindo Pharma USA, Inc. It includes dosing instructions, storage conditions, and precautions. The recommended dosage can be found in the package insert. The tablets should be stored at 20° to 25°C and should not be crushed, cut, or chewed. It advises against using the tablets if the inner seal of the bottle is broken or missing. The code mentioned is TS/DRUGS/24/2015. Overall, it is essential to follow the provided guidelines for the safe and effective use of this medication.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.