Atorvastatin Calcium Tablet, Film Coated
Product Images NDC 59651-607
View Photos of Packaging, Regulatory Labels, and Product Appearance
Product Visual Gallery
This gallery contains 8 technical images submitted to the FDA as part of the official labeling for Atorvastatin Calcium (NDC 59651-607). Unlike standard consumer photos, these assets often include clinical data figures, molecular chemical structures, and official manufacturer packaging layouts.
As provided by Aurobindo Pharma Limited, these visuals offer a comprehensive scientific overview of the product's physical and chemical identity, aiding pharmacists and researchers in product verification and study.
Product Images & Figures Index
Figure (Atorvastatin Fig2)

This text provides information on the cumulative hazard percentages for Placebo and Atorvastatin treatments over a period of four years. The Hazard Ratio (HR) for Atorvastatin compared to Placebo is reported as 0.63 with a confidence interval of (0.48-0.83) and a p-value of 0.001. This data suggests a significant difference in the risk of experiencing the primary endpoint between the two treatments.*
Figure (Atorvastatin Fig3)

This text appears to be a statistical result related to a study or clinical trial evaluating the effect of Atorvastatin at different doses (10 mg and 80 mg) on the time to first major cardiovascular endpoint over a certain period of time. The hazard ratio (HR) indicates a reduction in risk for the higher dose group compared to the lower dose group.*
Package Label-principal Display Panel (10 mg (90 Tablet Bottle))

This is a description for a bottle of Atorvastatin Calcium tablets (10 mg) manufactured by AUROBINDO. The prescription-only medication comes in a packaging of 90 tablets. The dosage and usage information can be found in the package insert. The tablets should be stored at a temperature range of 20° to 25°C. Distributed by Aurobindo Pharma USA, Inc. with the manufacturing taking place in India. The product code is TS/DRUGS/22/2009.*
Package Label-principal Display Panel (20 mg (90 Tablet Bottle))

This is a description of Atorvastatin Calcium tablets, a prescription medication with a strength of 20 mg. The tablets are film-coated and packed in a bottle containing 90 tablets. The medication is distributed by Aurobindo Pharma USA, Inc. and made in India. The recommended storage temperature is between 20° to 25°C. Detailed prescribing information can be found in the package insert.*
Package Label-principal Display Panel (40 mg (90 Tablet Bottle))

This is a description of a medication called Atorvastatin Calcium in tablet form. Each film-coated tablet contains 40 mg of Atorvastatin Calcium USP. It is distributed by Aurobindo Pharma USA, Inc. and made in India. The tablets are to be stored at 20°C to 25°C and dispensed in tight containers. For full prescribing information, refer to the package insert.*
Package Label-principal Display Panel (80 mg (90 Tablet Bottle))

This is a description for Atorvastatin Calcium Tablets, USP. Aurobindo Pharma USA, Inc. distributes these tablets containing 80 mg of atorvastatin each. The product comes in a pack of 90 tablets and is for prescription use only. It is advised to store these tablets at a temperature between 20°C to 25°C. For detailed dosage and usage instructions, refer to the package insert. This medication is manufactured in India.*
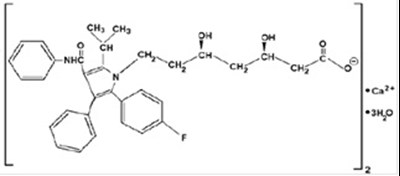
Str (Atorvastatin Str)
* These product label images have been analyzed using experimental machine learning. Please verify findings with the primary label text.