Product Images Esomeprazole Magnesium
View Photos of Packaging, Labels & Appearance
- figure - esomeprazole fig1
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 40 mg (30 Packets Carton) - esomeprazole fig10
- figure - esomeprazole fig2
- figure - esomeprazole fig3
- figure - esomeprazole fig4
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg (Packet) - esomeprazole fig5
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg (30 Packets Carton) - esomeprazole fig6
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 mg (Packet) - esomeprazole fig7
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 20 mg (30 Packets Carton) - esomeprazole fig8
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 40 mg (Packet) - esomeprazole fig9
- str - esomeprazole str
Product Label Images
The following 11 images provide visual information about the product associated with Esomeprazole Magnesium NDC 59651-802 by Aurobindo Pharma Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 40 mg (30 Packets Carton) - esomeprazole fig10

This is a product description for Esomeprazole Magnesium Delayed-Release Oral Suspension. The text contains information about the NDC number (59651-805-30) and instructions for dispensing the medication guide to each patient. It also mentions the manufacturer, Aurobindo, and the packaging details.*
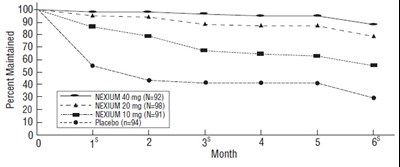
figure - esomeprazole fig3
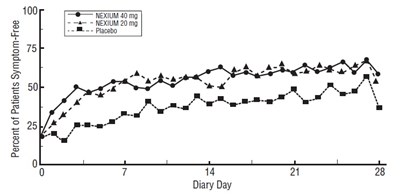
The text provides information about the percent of patients who were symptom-free after taking NEXIUM 40 mg and NEXIUM 20 mg on day 14 of the Diary.*
figure - esomeprazole fig4

This text shows data on the percentage of patients who were symptom-free while taking Nexium in two different doses (40 mg and 20 mg) compared to a placebo. The information is recorded for various diary days.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 5 mg (30 Packets Carton) - esomeprazole fig6

This text appears to be a summary of a medication prescription for Esomeprazole Magnesium in delayed-release oral suspension. The medication is available in single-dose packets and should be dispensed with a Medication Guide for each patient. The prescription is labeled for Rx only.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.