Product Images Valsartan
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 14 images provide visual information about the product associated with Valsartan NDC 59746-362 by Jubilant Cadista Pharmacuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
160mgcarton

The text describes a medication called Valsartan. The package has been manufactured in India by Jubilant Generics Ltd and Jubilant Cadista Pharmaceuticals Inc. The contents of the package are 100 tablets with each film-coated tablet containing Valsartan USP 160 mg. According to the instructions, the usual dosage can be found in the accompanying prescribing information. The tablet package warns that it is important to keep all medications out of reach of children and to store them away from moisture. If the package is dispensed for outpatient use, a child-resistant container should be used. There is an apparent reference to an NDC code, but no additional actionable or readable information is available.*
160mgfoil

The text is a label for Valsartan tablets, with instructions to use only as marked by B, lot number and expiration date provided. The tablets meet USP standards and are for prescription use only.*
160mglabel

This appears to be a medication label that contains information about a drug called Valsartan. It has a suggested dosage and instructions to keep it out of reach of children. This medication should be stored at room temperature and protected from moisture. The label indicates that it contains 90 tablets and includes information about the manufacturer.*
320mgcarton

This is a description of a medication package containing Valsartan Tablets USP 320 mg. The package includes 30 tablets and is manufactured by Jubilant Generics Ltd and Jubilant Cadista Pharmaceuticals. It includes information about dosage and warnings to keep the medication away from children, store at a certain temperature, and protect from moisture. The package is not child-resistant, and a child-resistant container should be used for dispensed outpatient use. There is also a barcode on the package with the numbers "26-69€-9v.63 OON."*
320mgfoil

This is a not-available description as the text appears to be a combination of different characters and numbers without any discernible meaning or context.*
320mglabel

This is a medication called Valsartan in the form of film-coated tablets, with a strength of 320 mg per tablet. The recommended dosage and other relevant information are provided in the prescribing information that accompanies the medication. It is advised to keep the medication out of reach of children, protect it from moisture, and store it in a tight container in a temperature-controlled environment. The medication is manufactured by Jubilant Generica Ltd in India and marketed by Jubilant Cadista Pharmaceutcals in the USA. The NDC code for this medication is 59746-363-90, and it comes in a bottle containing 90 tablets. There is a warning to dispense it along with patient information sheet.*
40mgcarton

This is a description of Valsartan Tablets USP 40mg manufactured by Jubilent Generics Ltd. The tablets are used to treat high blood pressure and each film-coated tablet contains Valsartan USP 40mg. The usual dosage needs to be determined while referring to the accompanying prescribing information. The tablets are packed as 100 tablets in a package, which is not child-resistant. It is advised to keep this and all medication out of reach of children and store at 20° to 25°C.*
40mgfoil

This is a collection of data that pertains to Valsartan Tablets marketed by Jubiant Cadista Pharmaceuticals. The tablets are of the USP 40mg variety and are made and/or manufactured in India. The data also includes Lot and Exp numbers.*
40mglabel
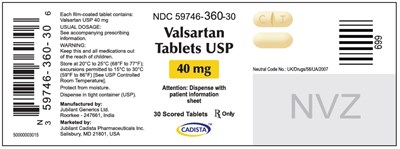
The text is a medication label for Valsartan, which is available in tablet form containing 40 mg of the drug. The usual dosage is provided in accompanying prescribing information. The label warns that the medication should be kept away from children, protected from moisture and stored at a temperature of 20°C to 25°C. The medication is manufactured by Jubllant Generics Ltd in India and marketed by Jubllant Cadista Pharmaceuticals Inc in Salisbury, MD. The NDC number for this medication is 59746-360-30.*
80mgfoil

This text provides information related to the product Valsartan Tablet USP. It includes the name of the manufacturer, Jubilant Cadista Pharmaceuticals Inc., and the product code 59746-361-03 7. The tablets are marked by R Only, and the product is made in an unspecified location. The text also indicates that the tablets are available in different dosages.*
80mglabel

This is a medication called Valsartan, which comes in film-coated tablets of 80 mg. The recommended dosage is not provided and it's advised to consult the prescribing information. The medicine should be kept out of reach of children and stored between 20°C to 25°C (68°F to 77°F) with excursions permitted to 15°C to 30°C (59°F to 86°F). It should also be protected from moisture and dispensed in a tight container. The manufacturer is Jubilant Generics Ltd. located in Roorkee, India and it's marketed by Jubilant Cadista Pharmaceuticals Inc. in Salisbury, MD, USA. The neutral code number is mentioned as UKIDIugs/S81UAI2007.*
fig01
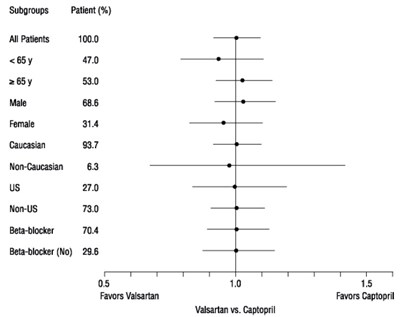
This appears to be a table showing patient subgroups and results of a study comparing the effectiveness of Valsartan versus Captopril. The subgroups shown include age, gender, race, US residency, and use of beta-blockers. The results are expressed as percentages favoring Valsartan or Captopril within each subgroup. However, the table is not easily readable due to formatting issues, making it difficult to provide a more detailed description.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

