Product Images Sildenafil
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 26 images provide visual information about the product associated with Sildenafil NDC 59762-0033 by Greenstone Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 7 - sildenafil 08

This document appears to be a chart or a table showing the recommended dosage adjustments for various drugs when taken in combination with sildenafil, based on the drug's AUC and Cmax values. The chart indicates that most of the drugs listed do not require any dose adjustment when taken with sildenafil, however, coadministration of sildenafil with Azithromycin, Ritonavir, and Saquinavir is not recommended. The document also includes a graph showing the change relative to Sildenafil alone.*
Figure 8 - sildenafil 09

This text appears to be a list of drugs (including Acenocoumarol, Aspirin, Atorvastatin, Bosentan, etc.) and their interactions with other drugs (such as Ritonavir and saquinavir), as well as the recommended dosage adjustments. It also includes information on the impact of the drugs on INR AUC, Cmax, and BT, as well as the Fold Change and 90% CI. The text suggests that Doxazosint at certain levels interacts with 25mg, 50mg, or 100mg sildenafil. Finally, the text mentions that the addition of sildenafil to bosentan therapy does not benefit exercise capacity.*
Figure 9 - sildenafil 10

This appears to be a table showing changes in measurements (possibly distances) between two locations labeled "Placebo" and "Sildenafil" under various dosages ("40mg TID", "80mg TID", "20mg TID"). It is a medical document, likely related to a study on the effects of sildenafil on patients.*
Figure 10 - sildenafil 11

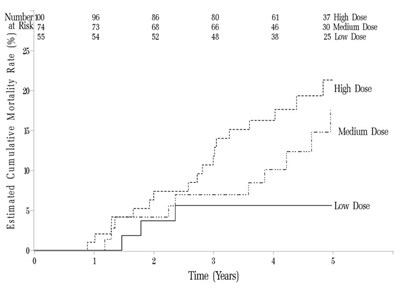
Placobo Sidenatl & Sidonati is a medication that is prescribed to patients with pulmonary arterial hypertension (PAH) who have functional capacity issues. The text also includes some numbers that may relate to patient testing or other medical indicators, such as "Basalino walk distance" and "dynesseciomin?" However, some of the text is not readable or in a different language, and without more context it is difficult to determine the usefulness of this description.*
Figure 12 - sildenafil 13

The text describes a plot showing the mean change from baseline in meters with standard errors for each treatment at each visit up to week 12. The treatments include Sildenafil ImgTID, Sildenafil 5mg TD, and Sildenafil 20mg TID. The plot has data for Week 4, Week 8, and Week 12.*
Logo - sildenafil 14

The Greenstone brand is distributed by Greenstone LLC, located in Peapack, NJ 07977.*
Logo - sildenafil 15

This is the packaging information of the Greenstone® brand, distributed by Greenstone LLC from Peapack in NJ, USA.*
Logo - sildenafil 23

The Greenstone brand is distributed by Greenstone LLC located in Peapack, NJ 07977.*
PRINCIPAL DISPLAY PANEL - 20 mg Tablet Bottle Label - sildenafil 24

Store the medication at a controlled room temperature ranging from 20°C to 25°C (B8°F to 77°F) with permitted excursions to 15°C to 30°C (BO°F-6°F) as per USP controlled room temperature. Dispense the medication in tight, child-resistant containers. Refer accompanying prescribing information for dosage and use guidance. Each tablet contains 1020 mg sildenafil equivalent. Distributed by Greenstone (LC Peapack, NJ 07977). The NDC number is 59762-0033-1, and it is available in a bottle of 90 tablets under the Greenstone brand.*
PRINCIPAL DISPLAY PANEL - 10 mg/mL Bottle Label - sildenafil 25

This is a medication for oral use called Sildenafil, available as a greenstone brand. It is to be stored below 30°C in its original package to protect from moisture before reconstitution. After reconstitution, it can be stored below 30°C or in a refrigerator at 2°C to 8°C. It should not be frozen or used after 60 days of reconstitution. Each milliliter of the suspension contains 10mg of Sildenafil and it is recommended to shake well before every use. The medication is available in Grape Flavor and it is intended for oral use only. Recommended dosage instructions are not available.*
PRINCIPAL DISPLAY PANEL - 10 mg/mL Bottle Carton - sildenafil 26

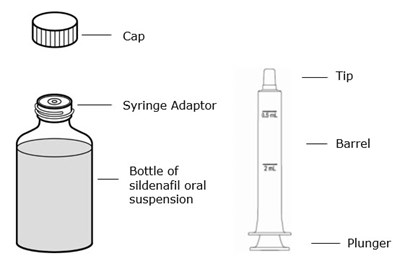
This is a product description for Greenstone brand sildenafil for oral suspension. The suspension has a grape flavor and contains sildenafil at a concentration of 10 mg/mL. The product comes as a powder and must be reconstituted with water before use. Each bottle contains 112 mL of suspension, which must be stored below 30°C or in the refrigerator. The recommended dosage is not provided in the description. The product also includes directions for reconstitution and includes a dosing syringe and press-in bottle adapter. Greenstone LLC, located in Peapack, NJ, is the manufacturer and the National Drug Code (NDC) is 59762-2056-1.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.