Product Images Desvenlafaxine Succinate
View Photos of Packaging, Labels & Appearance
- Chemical Structure - desvenlafaxine 01
- Figure 1 - desvenlafaxine 02
- Figure 2 - desvenlafaxine 03
- Figure 3 - desvenlafaxine 04
- Figure 4 - desvenlafaxine 05
- Figure 5 - desvenlafaxine 06
- Logo - desvenlafaxine 07
- Logo - desvenlafaxine 08
- PRINCIPAL DISPLAY PANEL - 50 mg Tablet Bottle Label - desvenlafaxine 09
- PRINCIPAL DISPLAY PANEL - 100 mg Tablet Bottle Label - desvenlafaxine 10
- PRINCIPAL DISPLAY PANEL - 25 mg Tablet Bottle Label - desvenlafaxine 11
Product Label Images
The following 11 images provide visual information about the product associated with Desvenlafaxine Succinate NDC 59762-1211 by Greenstone Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - desvenlafaxine 02
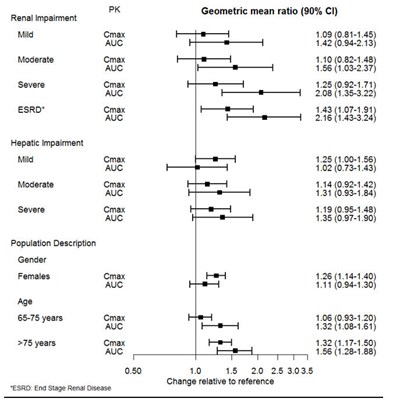
This text seems to be a chart or table related to the pharmacokinetics of a drug in different populations with varying degrees of renal and hepatic function, as well as gender and age. It shows values of Cmax and AUC (maximum concentration and area under the curve) for each group. However, it is not possible to determine the specific drug or dosage being evaluated, so further information is needed to interpret the results.*
Figure 2 - desvenlafaxine 03
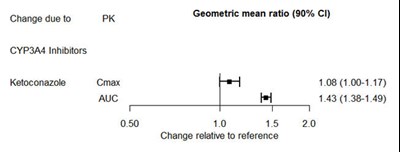
The text describes the effect of CYP3A4 inhibitors, particularly ketoconazole, on Cmax and AUC levels. The geometric mean ratios (with 90% confidence intervals) are provided for both Cmax and AUC values. The table shows the percent change relative to the reference value of 0.50 for different time intervals.*
Figure 4 - desvenlafaxine 05
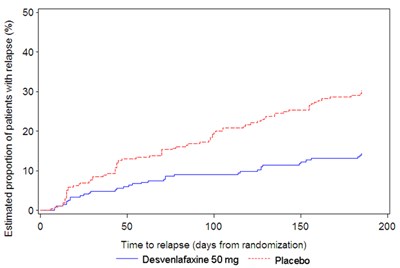
This is a chart or graph that shows the estimated proportion of patients with relapse over time for Desvenlafaxine 50 mg and Placebo. The y-axis shows the percentage of patients with relapse (ranging from 40% to 50%) while the x-axis shows the time to relapse (measured in days from randomization). There are three data points on the x-axis: 50, 100, and 150. The chart shows that Desvenlafaxine 50 mg has a lower proportion of patients with relapse compared to Placebo over time. However, without additional context, it is unclear what condition or study is being referred to.*
Figure 5 - desvenlafaxine 06
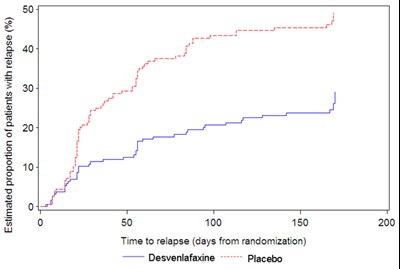
This text presents a graph showing the estimated proportion of patients with relapse over time (measured in days from randomization) for two groups: one that received Desvenlafaxine and another that received a placebo. The graph displays data points at intervals of 50 days up to 150 days. The y-axis measures the proportion of patients with relapse as a percentage, while the x-axis measures the time to relapse.*
Logo - desvenlafaxine 07

This text is a brief description of the company Greenstone LLC, which distributes a brand called "Greenstone" based in Peapack, NJ 07977.*
Logo - desvenlafaxine 08

This is a description of the Greenstone® brand, which is distributed by Greenstone LLC in Peapack, NJ 07977.*
PRINCIPAL DISPLAY PANEL - 50 mg Tablet Bottle Label - desvenlafaxine 09

Each table of this medication contains 76 mg of desvenlafaxine succinate, which is equivalent to 50 mg of desvenlaasine. The proper dosage should be obtained from the package insert. The medication should be stored between 20°C and 25°C (58°F to 86°F), with excursions permitted between 18°C and 30°C (64°F to 86°F). The medication is distributed by Greenstone LLC from Peapack, NJ and is made in Singapore. The package contains 30 tablets, and should always be dispensed with a medication guide. The package is intended to be dispensed as a unit.*
PRINCIPAL DISPLAY PANEL - 25 mg Tablet Bottle Label - desvenlafaxine 11

This is a medication description for Desvenlafaxine Succinate extended-release tablets. It contains 30 tablets with each tablet containing 38 mg of the medication. The medication guide for this drug should always be dispensed. The medication is manufactured by Greenstone LLC and is stored between 20° to 25°C (68° to 77°F). The medication is distributed in the United States and made in Singapore.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


