Product Images Triazolam
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Triazolam NDC 59762-3718 by Greenstone Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Logo - triazolam 02

The Greenstone Brand is distributed by Greenstone LLC based in Peapack, NJ 07977.*
Logo - triazolam 02a

This is a label for a brand called "Greenstone" and it is distributed by a company named "Greenstone LLC" located in Peapack, NJ 07977.*
PRINCIPAL DISPLAY PANEL - 0.125 mg Tablet Bottle Label - triazolam 03
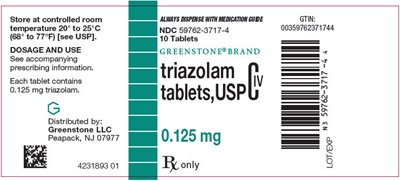
This is a description of a medication containing 0.125 mg of triazolam in each tablet. It should be stored at a controlled room temperature between 20 to 25°C (68° to 77°F) as per the USP guidelines. The medication is distributed by Greenstone LLC, and the NDC number is 59762-3717-4. The prescribing information should be consulted for dosage and use. The GTIN number is 0035976237174.*
PRINCIPAL DISPLAY PANEL - 0.25 mg Tablet Bottle Label - triazolam 04
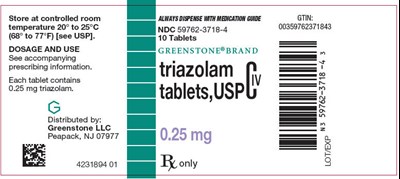
Greenstone® brand triazolam tablets (0.25 mg) should be stored at a controlled room temperature of 20° to 25°C (68° to 77°F). These tablets are available in a pack of 10 with NDC 50762-3718-4 and GTIN 00359762371843. The medication should always be dispensed with a medication guide. Dosage and usage instructions can be found in the accompanying information. This medication is distributed by Greenstone LLC located in Peapack, NJ 07977.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
