Product Images Sildenafil
View Photos of Packaging, Labels & Appearance
- 1 - sildenafil powder for oral suspension 1
- E - sildenafil powder for oral suspension 10
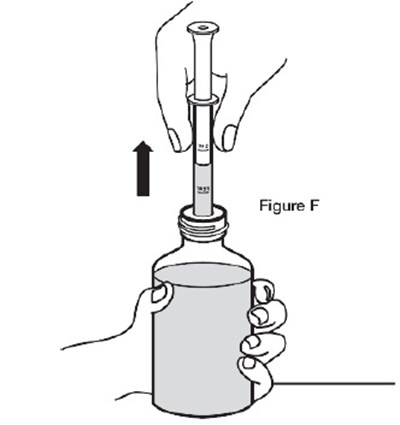
- F - sildenafil powder for oral suspension 11
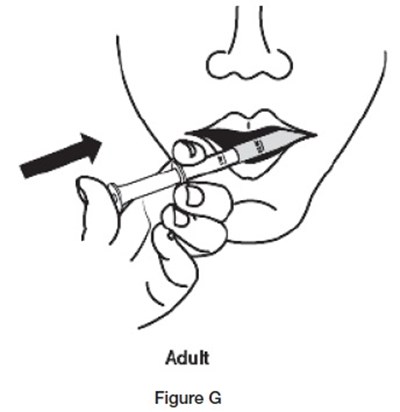
- G - sildenafil powder for oral suspension 12
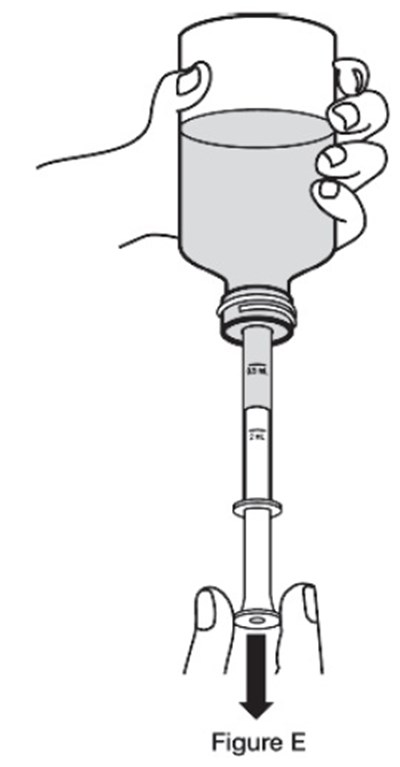
- H - sildenafil powder for oral suspension 13
- I - sildenafil powder for oral suspension 14
- 1 - sildenafil powder for oral suspension 15
- 2 - sildenafil powder for oral suspension 16
- 2 - sildenafil powder for oral suspension 2
- 3 - sildenafil powder for oral suspension 3
- 4 - sildenafil powder for oral suspension 4
- 5 - sildenafil powder for oral suspension 5
- A - sildenafil powder for oral suspension 6
- B - sildenafil powder for oral suspension 7
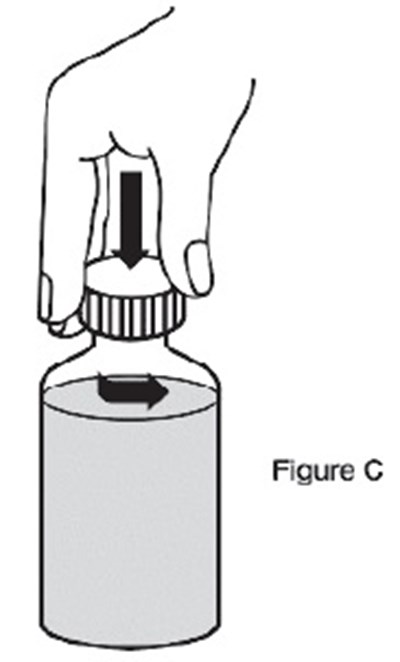
- C - sildenafil powder for oral suspension 8
- D - sildenafil powder for oral suspension 9
Product Label Images
The following 16 images provide visual information about the product associated with Sildenafil NDC 60219-2651 by Amneal Pharmaceuticals Ny Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
1 - sildenafil powder for oral suspension 1
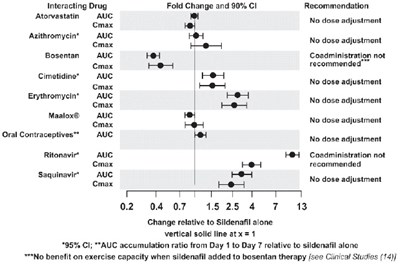
This text provides guidance on drug interactions with Atorvastatin, Bosentan, Cimetidine, Erythromycin, Maalox, Oral Contraceptives, Ritonavir, and Saquinavir. It indicates whether dose adjustment is needed for specific drugs when co-administered. The information includes data on fold change, 90% CI recommendations, and effects on AUC and Cmax values for various medications. It mentions that no benefit on exercise capacity is seen when adding sildenafil to bosentan therapy.*
1 - sildenafil powder for oral suspension 15

This text contains information about sildenafil suspension before reconstitution. It mentions that each bottle contains 1.57g of sildenafil citrate. The suspension should be stored below 30°C, protected from moisture, not be frozen, and any unused portion should be discarded after 60 days. The usual dosage should be followed as per the prescribing information. The manufacturer is Amneal Pharmaceuticals Pvt. Ltd. and the product is grape-flavored. The expiration date is listed as 02.2024-01.*
2 - sildenafil powder for oral suspension 16

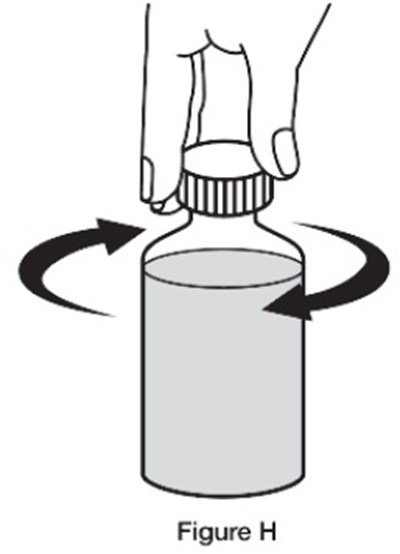
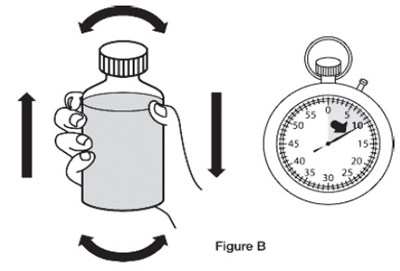
This text provides instructions for reconstituting a pharmaceutical suspension containing Sildenafil. It explains the process of adding water and shaking the bottle to ensure a uniform mixture. The recommended dosage and storage conditions are also mentioned. The product is specified for oral use only and each milliliter of the suspension contains 10 mg of Sildenafil. Manufacturing and distribution information is included at the end of the text.*
2 - sildenafil powder for oral suspension 2
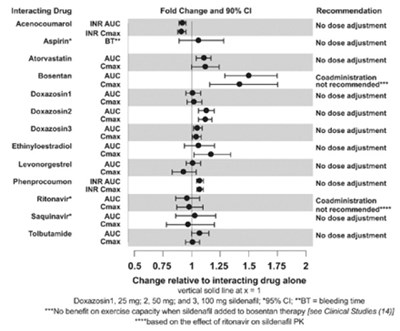
This text provides recommendations for drug interactions based on the fold change and 90% confidence intervals. It specifies advice for various drugs such as acenocoumarol, aspirin, atorvastatin, bosentan, doxazosin, ethinyl estradiol, and more. The text also includes specific adjustments or coadministration recommendations for different drugs.*
4 - sildenafil powder for oral suspension 4
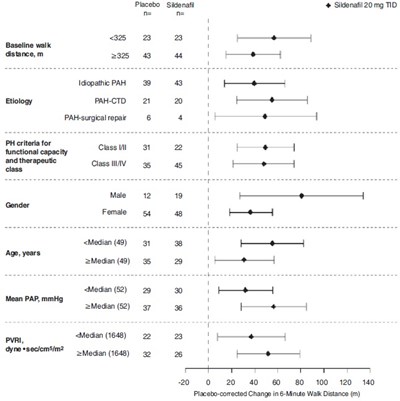
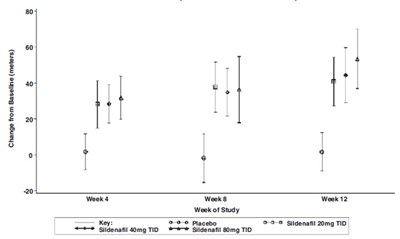
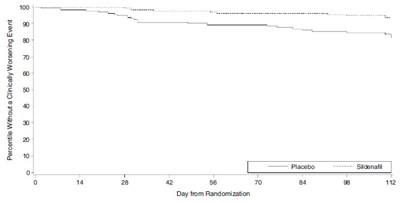
This text provides information on a study involving Placebo (Sildenafil) 20 mg taken three times a day for patients with pulmonary arterial hypertension (PAH). It includes median and mean values for various parameters such as age, pulmonary arterial pressure (PAP), and the 6-minute walk distance. The data is being presented for the placebo-corrected change in the 6-minute walk distance.*
A - sildenafil powder for oral suspension 6
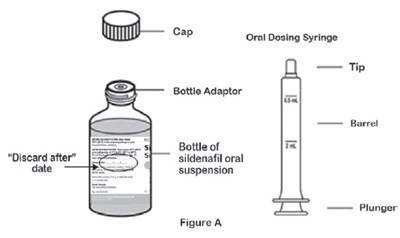
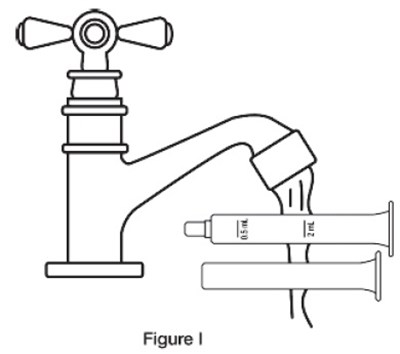
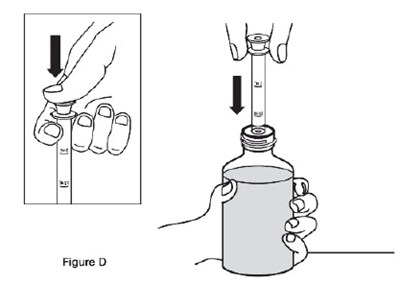
This text appears to describe different components of a bottle adapter and barrel used for a sildenafil oral suspension. It mentions a plunger and provides a reference to Figure A for visual guidance.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.