Product Images Paroxetine Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Paroxetine Hydrochloride NDC 60505-1318 by Apotex Corp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Apo Paxil CR12 5mg

Each extended-release tablet contains 12.5 mg of paroxetine hydrochloride. It is important to store the product at or below 25°C (77°F) and use safety closures when dispensing the product, unless otherwise directed by a physician or requested by the purchaser. The medication guide provided with the bottle should be used when dispensing the product. The manufacturer is Apotex, located in Toronto, Canada, and the product is distributed in the United States by Apotex Corp. The NDC number for this product is 60505-1316-3. Dosage information can be found in the accompanying prescribing information.*
Apo Paxil CR25mg

Each tablet contains 25 mg of paroxetine hydrochloride and is intended for extended release purposes. The tablets must be stored below 25°C and safety closures are recommended. The text also includes a product code, dosage and manufacturing details. The dispensing of these tablets is subject to federal law and a medication guide should be provided. The product is manufactured by Apotex Corp. in Canada.*
Apo Paxil CR37 5mg

Each tablet contains 37.5 mg of paroxetine hydrochloride. The extended-release tablets should be stored below 25°C. The medication guide provided should be dispensed with the tablets. The prescription must be accompanied with the dosage information. Safety closures should be used when dispensing this medication unless directed otherwise by the physician or requested by the purchaser. The manufacturer is Apotex Corp. The product is marked with an NDC number and batch number.*
Figure1
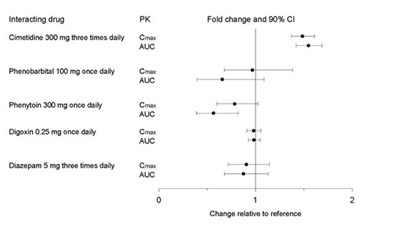
This text appears to be a list of various drugs with their dosages and a section labeled "PK" and "Fold change and 90% CI". It is not clear what the purpose of the list is or what the abbreviations and terms signify. Therefore, a useful description cannot be generated from this information.*
GSK Paxil CR12 5mg

This is a description of a medication named "Paroxetine HOI". It comes in tablet form with 30 tablets in each container. Each tablet contains 125mg of paroxetine hydrochloride. The NDC number for this medication is 60505-3673-3. The usual dosage instructions can be found in the package insert. The medication should be stored in a tightly closed and light-resistant container. The manufacturer of the medication is GlaxaSmithKline and it is distributed by Apotex Corp.*
GSK Paxil CR25mg

Each controlled-release tablet contains paroxetine hydrochloride equivalent to 25 mg paroxetine. It is recommended to store below 25°C (77°F) and dispense in a tight, light-resistant container. The usual dosage can be found in the package insert. Safety closures should be used when dispensing the product if not otherwise directed by a physician or requested by the purchaser. This product was manufactured by Apotex Corp. in Ireland and contains 30 tablets with NDC 60505-3674-3. Paroxetine HCI is the active ingredient, and it is a controlled substance for which a prescription is required.*
GSK Paxil CR37 5mg

Each controlled-release tablet contains 37.5 mg paroxetine hydrochloride. The tablets should be stored below 25°C (77°F) and dispensed in a tight, light-resistant container. The usual dosage should be followed as per package insert. It is important to use safety closures when dispensing this product unless otherwise directed by the physician or requested by purchaser. This product is manufactured in Ireland by GlaxoSmithKline for Apotex Corp. The NDC number for this product is 60505-3675-3.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.