Product Images Teriflunomide
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Teriflunomide NDC 60505-4477 by Apotex Corp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure1.jpg - Figure1
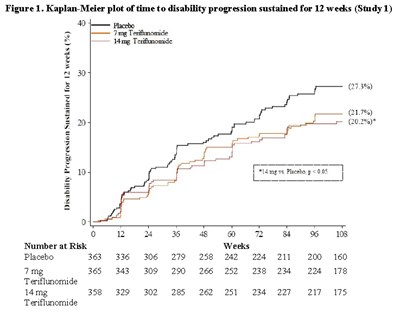
Figure 1 is a Kaplan-Meier plot illustrating the time to disability progression sustained for 12 weeks in Study 1. The plot includes data points representing the number at risk for the placebo group, the 7 mg Teriflunomide group, and the 14mg Teriflunomide group. The percentages indicate the progression rates in each group, with the placebo group showing a rate of 3%, the 7 mg Teriflunomide group at 17%, and the 14mg Teriflunomide group at 202%. The plot also shows the number of individuals at various time points.*
Figure2.jpg - Figure2
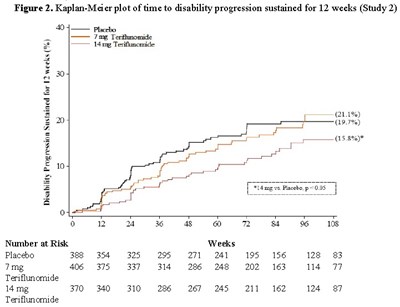
The text provides a Kaplan-Meier plot showing the time to disability progression sustained for 12 weeks in Study 2. The plot compares the results of three different treatments: placebo, 7mg Teriflunomide, and 14 mg Teriflunomide. The plot also shows the number of participants at risk at various weeks.*
bottle14mg.jpg - bottle14mg

This is a description of a medication called Teriflunomide tablets. Each tablet contains 14 mg of teriflunomide. The tablets should be stored at a temperature of 20°C - 25°C (68°F - 77°F) with excursions permitted to 15°C - 30°C (59°F - 86°F) according to USP Controlled Room Temperature guidelines. The medication should be dispensed in a tight container. The text includes a warning to keep the medication out of the reach of children. The suggested dosage and administration is one tablet to be taken orally once daily. The tablets can be taken with or without food. The packaging contains 30 tablets and includes an NDC number. The medication guide enclosed inside should be given to each patient by the pharmacist. The manufacturer of the tablets is Apotex Inc., based in Toronto, Ontario, Canada. The tablets are manufactured for Apotex Corp. located in Weston, Florida.*
bottle7mg.jpg - bottle7mg

This is a description of a medication called Teriflunomide. It is a film-coated tablet that contains 7 mg of the active ingredient. The tablets should be stored between 20°C - 25°C (68°F - 77°F), with excursions permitted to 15°C - 30°C (59°F - 86°F). The medication should be dispensed in a tight container and kept out of the reach of children. The recommended dosage is one tablet orally once daily. The tablets can be taken with or without food. The packaging contains 30 tablets with the NDC number 60505-4477-3. The medication is manufactured by Apotex Inc. in Toronto, Ontario, Canada, and distributed by Apotex Corp. in Weston, Florida. The pharmacist should dispense the enclosed Medication Guide to each patient.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


