Product Images Abiraterone Acetate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Abiraterone Acetate NDC 60505-4764 by Apotex Corp., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2 - 03
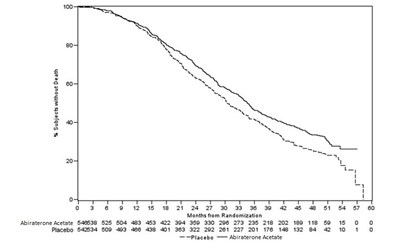
This appears to be a chart or graph showing the number of subjects without death over a period of time. The data compares the effectiveness of Abiraterone Acetate and a placebo. Abiraterone Acetate seems to have had higher survival rates than the placebo over time.*
Figure 3 - 04

The text seems to be a table showing the percentage of subjects without radiographic progression after receiving either Abiraterone Acetate or a Placebo. The table includes the number of subjects for each treatment group and the time frame of 24 months from randomization. The last row shows the percentage difference between subjects on Abiraterone Acetate and those on Placebo.*
PRINCIPAL DISPLAY PANEL - 500 mg Tablet Bottle Label - bottle

This is a medication for which the usual dosage is specified in the package insert. The medication contains abiraterone acetate and is manufactured by Qilu Pharmaceutical for Apotex Corp. The tablets should not be crushed or chewed and should be kept out of reach of children. Each film-coated tablet contains 500 mg of abiraterone acetate, USP. The available quantity is 60 film-coated tablets.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


