Product Images Pemetrexed
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 14 images provide visual information about the product associated with Pemetrexed NDC 60505-6066 by Apotex Corp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure2 - Figure2
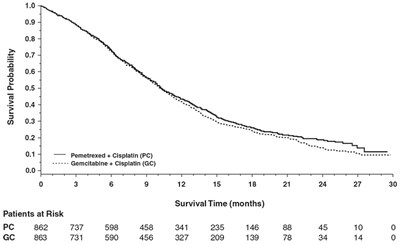
This is a table showing the survival probability of patients receiving either Pemetrexed + Cisplatin (PC) or Gemcitabine + Cisplatin (GC) treatments. The table also shows the number of patients at risk for each treatment and their survival times in months.*
Figure3 - Figure3
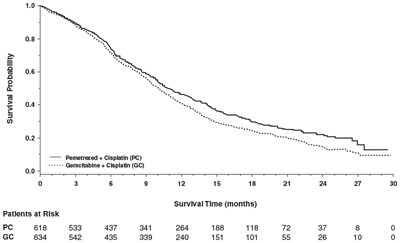
The text shows a chart representing the survival probability for two chemotherapy treatments, Pemetrexed + Cisplatin (PC) and Gemcitabine + Cisplatin (GC), over a period of 30 months. The chart shows the number of patients at risk for each treatment at different time intervals. The chart displays the data in months and the patient count for different time intervals.*
Figure4 - Figure4
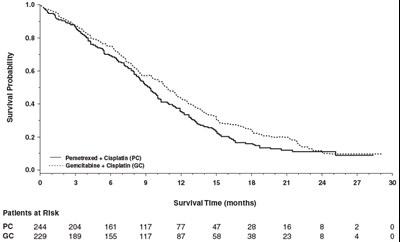
This appears to be a survival analysis table showing survival probability and time for two chemotherapy treatments (Pemetrexed + Cisplatin and Gemcitabine + Cisplatin) in patients with some type of cancer. The table includes the number of patients at risk and the number of patients who survived at various time lengths.*
Figure6 - Figure6
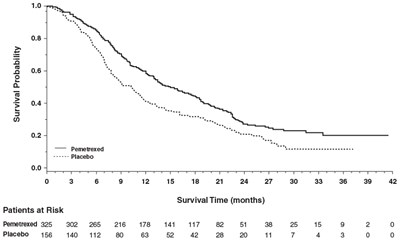
This seems to be a table showing survival probabilities and time for patients at risk, divided into groups receiving Pemetrexed and a Placebo. There are also some numerical values. However, without further context, it is impossible to determine what medical condition or study this data belongs to.*
Figure7 - Figure7
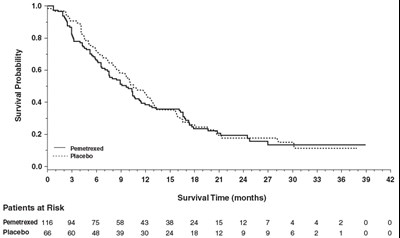
This is a chart representing the survival probability of patients who were either treated with Pemetrexed or Placebo. The graph shows the survival time in months for patients with corresponding patients at the risk values.*
Figure9 - Figure9
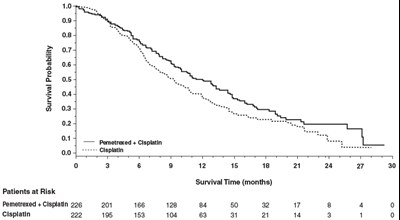
This is a graph showing Survival Probability based on the administration of Pemetrexed + Cisplatin versus Cisplatin alone over a period of 30 months. The graph presents the number of patients at risk for each drug regimen over time.*
CartonLabel100mg - Pemetrexed 100mg Carton

This appears to be a description of a medication called Pemetrexed. It comes in a single-dose vial with 100 mg/vial, and is intended for intravenous use only. The medication must be reconstituted by adding 4.2 ml of 0.9% Sodium Chloride Injection. The reconstituted solution must be used within 24 hours, and any unused portion must be discarded. Storage instructions are also included.*
CartonLabel500mg - Pemetrexed 500mg Carton
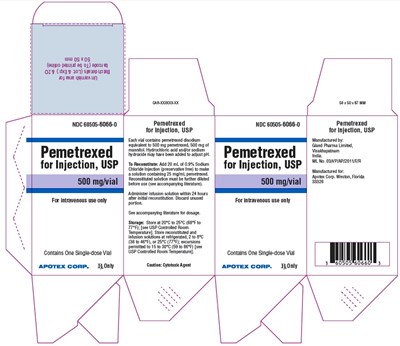
This is a description of a drug for intravenous use only. The drug is called Pemetrexed and the manufacturer is Apotex Corp. The drug is available as a 500 mg/vial single-dose vial. The drug needs to be reconstituted with 20 mL of 0% sodium chloride injection, which will result in a solution containing 25 mg/mL of Pemetrexed. This solution should be used within 24 hours of reconstitution. The drug should be stored at 20°C to 5°C in a USP controlled room temperature. The text also provides a cautionary note about the drug being a cytotoxic agent. The accompanying literature provides information about the dosage and administration of the drug.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.





