Product Images Rivaroxaban
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Rivaroxaban NDC 60505-6256 by Apotex Corp., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
btl-lbl-500s.jpg - btl lbl 500s

This information pertains to Rivaroxaban tablets, USP containing 25 mg of the active ingredient. The storage conditions recommend keeping the tablets at a temperature between 20°C to 25°C. The medication should be kept out of reach of children. The dosage information can be found in the package insert. The tablets are manufactured by Apotex Inc. for Apotex Corp. in Canada and the Medication Guide is available for patients.*
btl-lbl-60s.jpg - btl lbl 60s

This text contains information about a medication called rivaroxaban. Each film-coated tablet contains 2.5 mg of rivaroxaban, USP. It should be stored at 20°C to 25°C (68°F to 77°F) and should be kept out of reach of children. The usual dosage information can be found in the package insert. The medication is available in a pack of 60 tablets with the NDC 60505-6256-6. The tablets are manufactured by Apotex Inc. in Toronto, Ontario, Canada, and manufactured for Apotex Corp in Weston, Florida. Pharmacists are advised to dispense the medication guide available at the provided link to each patient.*
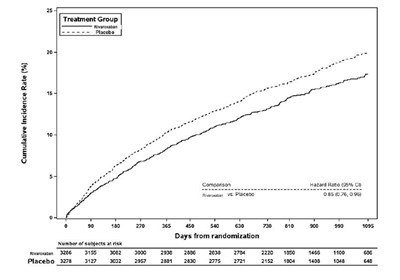
figure10.jpg - figure10

This text provides information regarding a study comparing Rivaroxaban to Placebo in terms of cumulative incidence rate and hazard ratio. The hazard ratio of 0.74 (95% CI: 0.65-0.86) indicates a lower risk associated with Rivaroxaban compared to Placebo. The data also shows the number of subjects at risk over different time intervals from randomization, illustrating the progression of the study over the days.*
figure2.jpg - figure2

This text provides a population description related to factors such as end-stage renal disease, renal impairment, age ranges, body weight categories, hepatic impairment, and pharmacokinetic parameters like Cmax and AuC in different subgroups. It includes comparisons such as fold change and 90% confidence intervals for various conditions like severe renal impairment, moderate renal impairment, and mid-range renal function. The text also mentions details about the subjects studied, including those with creatinine clearance levels in specific ranges.*
figure3.jpg - figure3

This text provides information on the Drug PK Fold Change and 90% CI values for interacting drugs in the presence of combined P-gp and Strong CYP3A Inhibitors like Ketoconazole and Ritonavir. It also includes values for Clarithromycin, Erythromycin, and Fluconazole under different inhibitory categories. Additionally, data for Rifampicin as a Strong CYP3A Inducer and for other drugs like Aspirin, Atorvastatin, Clopidogrel, Digoxin, Enoxaparin, Mazlox, Midazolam, Naproxen, Omeprazole, Ranitidine, and Warfarin are provided. The information seems focused on assessing the impact of various drugs on absorption and metabolism pathways.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.