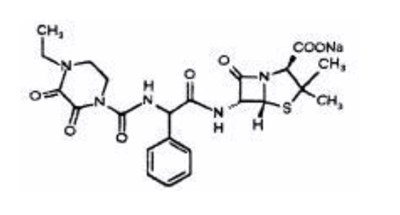
Product Images Piperacillin And Tazobactam
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Piperacillin And Tazobactam NDC 60505-6262 by Apotex Corp., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL – 40.5 grams Carton - carton label

This medication is labeled as Piperacillin and Tazobactam for Injection, USP and contains 36 grams of piperacillin and 4.5 grams of tazobactam with sodium. It requires reconstitution with 152 mL of a diluent to achieve a concentration of 200 mg/mL of piperacillin. The reconstituted solution should not be frozen, and any unused portion must be discarded after 24 hours at room temperature or after 48 hours when refrigerated. It is manufactured by Shandong Anxin Pharmaceutical Co., Ltd. and is intended for pharmacy bulk packages and not for direct infusion. Complete directions for use are available in the package insert.*
PRINCIPAL DISPLAY PANEL – 40.5 grams Vial - vial label

This is a bulk pharmacy package for intravenous injection containing piperacillin and tazobactam in powder form. It has to be reconstituted with a suitable diluent to form a solution of certain concentration before use. The solution, once made, should be used within 24 hours if stored at room temperature and 48 hours if refrigerated. The package insert has to be referred to for complete instructions for use. This package should not be directly infused and does not contain preservatives. The manufacturer is Apotex Corp., and the product is manufactured in China.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.