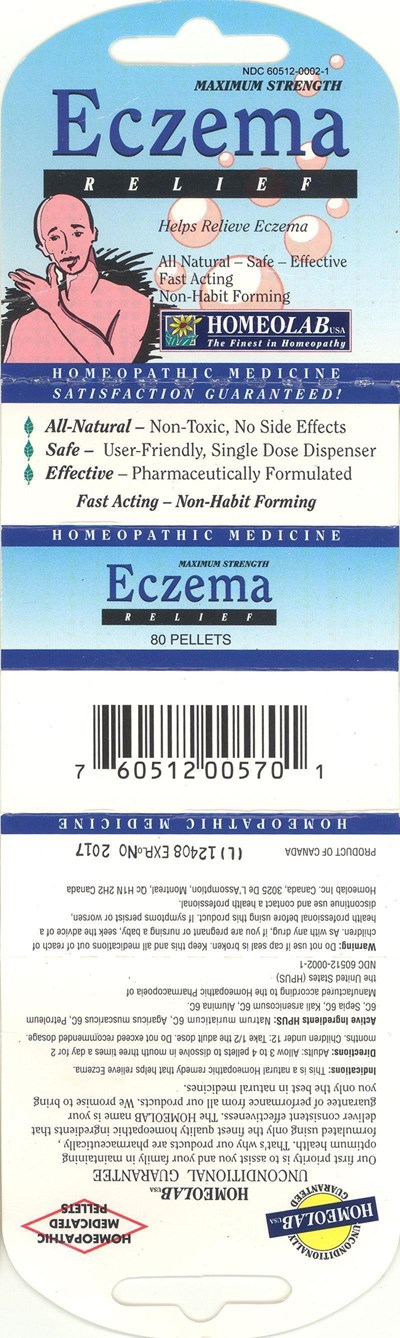
FDA Label for Eczema Relief
View Indications, Usage & Precautions
Eczema Relief Product Label
The following document was submitted to the FDA by the labeler of this product Homeolab Usa Inc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredients Hpus
Natrum muriaticum 6C
Agaricus muscarius 6C
Petroleum 6C
Sepia 6C
Kali arsenicosum 6C
Alumina 6C
References
Manufactured according to the Homeopathic Pharmacopoeia of the United States (HPUS)
Purpose
Eczema
Uses
This is a natural Homeopathic remedy that relieves:
- eczema
Otc - Ask Doctor
If symptoms persist or worsen, discontinue use and contact a health professional.
Otc - Pregnancy Or Breast Feeding
As with any drug, if you are pregnant or nursing a baby, seek the advice of a health professional before using this product.
Otc - Keep Out Of Reach Of Children
Keep this and all medications out of reach of children.
Directions
Adults: Allow 3 to 4 pellets to dissolve in mouth three times a day for 2 months.
Children under 12: Take 1/2 the adult dose.
Do not exceed recommended dosage.
Other Information
Do not use if cap seal is broken.
Homeolab USA Inc., 3025 De L'Assomption Blvd., Montreal, QC H1N 2H2 Canada
PRODUCT OF CANADA
Inactive Ingredients
Lactose, sucrose
Carton
* Please review the disclaimer below.