Product Images Aripiprazole
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 25 images provide visual information about the product associated with Aripiprazole NDC 60687-191 by American Health Packaging, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10 mg Aripiprazole Tablets Carton

This is the label information for a medication called Aripiprazole. The medication comes in tablet form, with 100 tablets in each bottle. Each tablet contains 10mg of Aripiprazole USP. The usual dosage amount is unclear, but it is recommended to read the package insert for full prescribing information. The medication should be stored at a controlled room temperature between 20-25°C (68-77°F), with slight excursions permitted between 15-30°C (59-86°F). The medication should be kept out of reach of children and the blister should not be used if it is torn or broken. The manufacturer is Gamber Pharmaceuticals and the medication is distributed by American Health Packaging.*
15 mg Aripiprazole Tablet Blister

This appears to be a list of medication names and dosages. The medications mentioned include Aripiprazole, and the dosages mentioned are 15mg. There are also some symbols and characters that are not clear.*
15 mg Aripiprazole Tablets Carton

This is a description of a medication called Aripiprazole in tablet form. It comes in a bottle of 30 tablets with a dosage of 15 mg each. The tablets should be stored between 3 to 20°C. The packaging information also includes a warning to keep the medication out of reach of children and to not use it if the blister pack is broken. The medication is distributed by American Health Packaging and comes from the manufacturer Cambor Pharmaceuticas. No additional information is available.*
2 mg Aripiprazole Tablet Blister

Not available. This text appears to be a combination of random letters, numbers, and symbols that do not form coherent sentences or phrases. It may have been incorrectly recognized by the software.*
2 mg Aripiprazole Tablets Carton

This is a description of a medication called Aripiprazole in tablet form. The medication comes in packages of 30 tablets and each tablet contains 2mg of Aripiprazole USP as the active ingredient. It is used for prescription-only purposes and should be dispensed with the accompanying medication. The medication should be stored in an environment between 20-25°C, and kept out of reach of children. The medication should not be used if the packaging is torn or broken. The manufacturer is American Health Packaging, and the NDC number is 60687-157-21.*
20 mg Aripiprazole Tablets Carton

This is a description of a medication called Aripiprazole in tablet form. The package contains 30 tablets with each tablet containing 20mg of Aripiprazole USP. The package has a National Drug Code (NDC) number of 60687-202-21. The medication should be stored at temperatures between 20°C to 25°C (68°F to 77°F) but excursions are permitted between 15°C to 30°C (59°F to 86°F). The package is distributed by Smerizan Fasit Packaging located in Columbus, Ohio and is for prescription use only. The package insert should be referred for full prescribing information and the medication should be kept out of reach of children.*
30 mg Aripiprazole Tablets Carton

This is a description of a medication that is identified by its NDC code and brand name, Aripiprazole. The medication comes as Tablets, USP with 30 Tablets (3x10) as a prescribed dosage. The package insert or full prescription information should be reviewed for the usual dosage. The tablets should be stored within a certain temperature range and must not be used if the blister is torn or broken. The medication is manufactured by Gamber Pharmaceutical, Inc. and distributed by American Health Packaging in Columbus, Ohio.*
5 mg Aripiprazole Tablets Carton

This is a description of a medication called Aripiprazole in tablet form. Each package contains 100 tablets, with a dosage of 5mg per tablet. The tablets are meant for prescription only and should be dispensed with a medication guide for patients by a pharmacist. The drug should be stored at room temperature and kept away from children. The package is distributed by American Health Packaging and has a National Drug Code (NDC) number of 60687-168-01.*
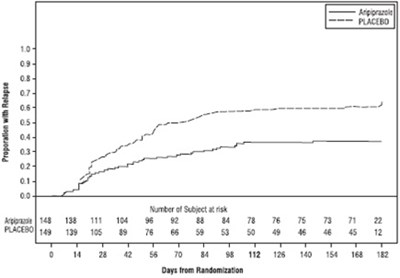
Figure1 - Figure1

The text provides information on the effect of different drugs on Aripiprazole. It includes details of inhibitors and inducers of various enzymes including CYP3A4 and CYP2D6, in addition to gastric acid blockers and miscellaneous drugs like valproate and lithium which can affect Aripiprazole. The data is organized in a table with fold change and 90% C1 information.*
Figure2 - Figure2

This text provides information on the effect of different drugs on Aripiprazole, a medication used to treat mental disorders such as schizophrenia and bipolar disorder. The text lists various drugs that interact with Aripiprazole, including inhibitors and inducers of enzymes responsible for metabolizing Aripiprazole in the body, such as CYP3A4 and CYP2D6. The text also mentions the effect of drugs that reduce gastric acid and other medications such as valproate and lorazepam. Additionally, the text includes a graph showing the fold change in Dehydro-Aripiprazole levels relative to a reference without interacting drugs, with different drug concentrations plotted on the x-axis.*
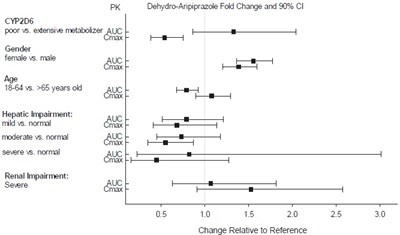
Figure4 - Figure4

This text appears to be a list of different factors that can affect drug metabolism and the analysis of drug levels in different populations. The text mentions PK (pharmacokinetics) and CYP206, which are both important factors in drug metabolism. It also compares the AUC and maximum levels of drugs in different populations based on gender, age, and hepatic impairment. The text also briefly mentions renal impairment and the impact of severe impairment on drug levels. The last line appears to indicate a graph showing the fold change and 0% C in avripiprazole, a particular drug, relative to a reference point.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.