Product Images Clozapine
View Photos of Packaging, Labels & Appearance
- 100 mg Clozapine Tablet Blister - 100 mg Blister
- 100 mg Clozapine Tablets Carton - 100 mg Carton
- 200 mg Clozapine Tablet Blister - 200 mg Blister
- 200 mg Clozapine Tablets Carton - 200 mg Carton
- 25 mg Clozapine Tablet Blister - 25 mg Blister
- 25 mg Clozapine Tablets Carton - 25 mg Carton
- 50 mg Clozapine Tablet Blister - 50 mg Blister
- 50 mg Clozapine Tablets Carton - 50 mg Carton
- AHP40401
- AHP41501
- AHP42601
- AHP54801
- Figure 1. Cumulative Probability of a Significant Suicide Attempt or Hospitalization to Prevent Suicide in Patients with Schizophrenia or Schizoaffective Disorder at High Risk of Suicidality - Figure 1
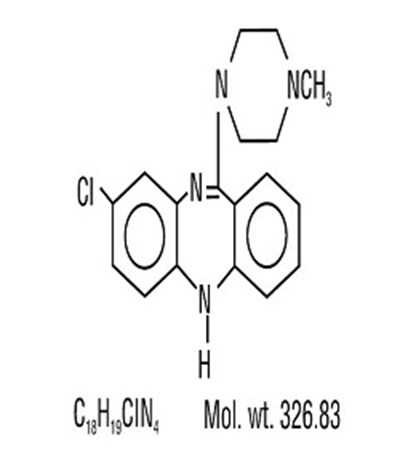
- Structural Formula - Structure
Product Label Images
The following 14 images provide visual information about the product associated with Clozapine NDC 60687-548 by American Health Packaging, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
100 mg Clozapine Tablets Carton - 100 mg Carton

NDC 60687-415-01 Clozapine Tablets USP is a prescription medication used for treating certain mental/mood disorders such as schizophrenia. Each package contains 100 tablets (10 x 10) of 100mg strength. The package mentions the recommended dosage for adults and storage instructions. The drug should be kept at a temperature of 20°-25°C, and kept away from children. The package also contains information about the manufacturer and distributor.*
200 mg Clozapine Tablets Carton - 200 mg Carton

This is a description of a package of Clozapine Tablets USP. The tablets are contained in a blister package of 100 tablets (10 x 10) and are only available through a prescription. Each tablet contains Clozapine USP. The description provides the usual adult dosage, Dispensing information, and storage details. It also includes a warning to keep the drug out of the reach of children. The package was manufactured by American Health Packaging and the drug process was contained in a package or form with NDC # 65852847 by Aurobindo Pharma USA, Inc.*
25 mg Clozapine Tablets Carton - 25 mg Carton

This is a medication package of Clozapine Tablets USP with an NDC code of 60687-404-01. Each package contains 100 tablets, with a potency of 25mg. The usual adult dosage of the drug is not mentioned, and the dispensing should be contingent upon ANC results. It should be kept out of reach of children and stored at 25-77°F. The medication is distributed by American Health Packaging from Ohio. The text contains a lot number, manufacturing date, and expiry date.*
50 mg Clozapine Tablet Blister - 50 mg Blister

The text is related to medication, specifically Clozapine tablets of 50 mg. The output also shows some product codes and batch numbers.*
50 mg Clozapine Tablets Carton - 50 mg Carton

Clozapine is a prescription drug and comes in the form of tablets. It contains 100 tablets in 10 strips of 10 each. Clozapine Tablets USP are recommended for adult use only and should be taken as per the prescription information provided along with the package. ANC test should be performed before dispensing the medicine. It should be stored at a temperature between 68°F to 77°F and protected from heat. This medicine should be kept out of reach of children. The product is distributed by American Health Packaging and the manufacturer is Aurabinda Pharma USA, Inc. The NDC number 60687-548-01 is also provided.*
Figure 1. Cumulative Probability of a Significant Suicide Attempt or Hospitalization to Prevent Suicide in Patients with Schizophrenia or Schizoaffective Disorder at High Risk of Suicidality - Figure 1

* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.