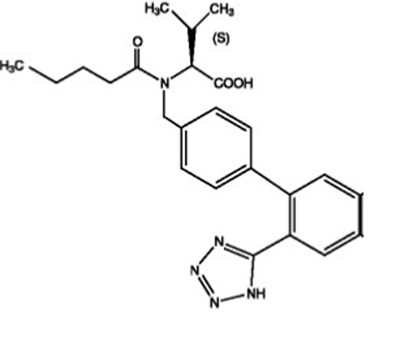
Product Images Valsartan
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Valsartan NDC 60687-612 by American Health Packaging, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
160 mg Valsartan Tablets Carton - 160 mg Carton

Valsartan Tablets USP is a prescription drug with NDC 60687-634-01. This package contains 100 tablets (10 x 10) of 60mg each. Usual Dosage instructions are available in the package insert. Store in the prescribed temperature range and protect from moisture. American Health Packaging is the distributor of this product manufactured by Ascend Laboratories, LLC.*
40 mg Valsartan Tablets Carton - 40 mg Carton

This is a description of a medicine called Valsartan in tablet form, with the National Drug Code (NDC) 60687-612-21. The package contains 30 tablets arranged in 3 groups of 10 tablets each, and the medicine is only available with a prescription. The tablets are USP compliant and contain Vasarian USP 0mg. The package insert provides instructions on the usual dosage. The medication needs to be stored at temperatures between 20 to 25°C, with excursions allowed between 15 to 30°C. The inner seal should protect the medication from moisture, and the package should be kept away from children. This product is distributed by American Hodih Packaging in Columbus, Ohio, and manufactured by Ascend Laboratories, LLC, with the NDC 60667-612-21.*
80 mg Valsartan Tablets Blister - 80 mg Blister

Valsartan is a medication commonly found in 80 mg dosage form with the product name "Yablar Up" by TaeiE. The package may include various lots and expiry dates. The manufacturer of this product is not identifiable from the provided text.*
80 mg Valsartan Tablets Carton - 80 mg Carton
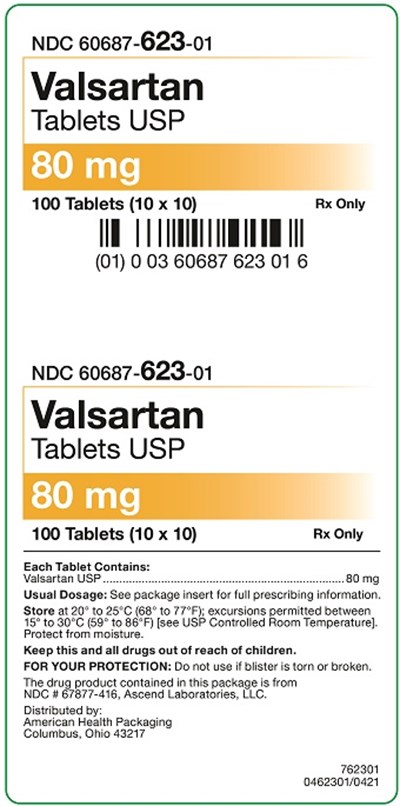
This is a description of a drug called Valsartan, which comes in the form of tablets that contains 50mg of Valsartan. The drug is packaged in a container that holds 100 tablets, and the recommended dosage can be found in the package insert for prescribing information. It is important to store the drug between 20-25°C with excursions permitted between 15-30°C, and to keep it out of reach of children. The drug is distributed by American Health Packaging in Ohio.*
Effects on Mortality Amongst Subgroups in VALIANT - Chart

This appears to be a table related to medical data or clinical trial data. It shows the percentage of patients in various subgroups, such as age, gender, race, location, and medication use, involved in a study or trial comparing the effects of Valsartan and Captopril. The last three lines indicate which treatment was favored by the study.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.