Product Images Duloxetine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 19 images provide visual information about the product associated with Duloxetine NDC 60687-723 by American Health Packaging, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
20 mg Duloxetine Delayed-Release Capsule Blister - 20 mg Blister

The text is a list of medications with their respective dosages and expiration dates. The medication listed is Duloxetine, available in a 20mg dose, and some capsules may contain delayed-release medicines. There are also different product LOTs provided.*
20 mg Duloxetine Delayed-Release Capsules Carton - 20 mg Carton

NDC 60687-723-21 is a drug package containing delayed-release capsules of Duloxetine USP. It is recommended to dispense with the accompanying medication guide for each patient. Each capsule contains 23 mg of duloxetine. The usual dosage should be reviewed using the package insert for complete prescribing information. The packaging should be kept in a controlled room temperature between 20 to 25°C, and it should be stored away from children. The package is distributed in Columbus, Ohio, by American Health Packaging. The capsules are equivalent to 20 mg of Forothlors USP. No other information is available.*
30 mg Duloxetine Delayed-Release Capsules Carton - 30 mg Carton

This is the description of a medication with the NDC code 60687-734-01. The medication is Duloxetine Delayed-Release Capsules USP that come in a package of 100 capsules (10 x 10) and it is only available with a prescription. The pharmacist is required to dispense this medication with its accompanying medication guide to each patient. Each capsule contains 337 mg of Duloxetine hydrochloride USP equivalent to 30mg duloxetine. The package insert should be referred to for full prescribing information. The medication should be stored between 15°C to 30°C (69°F to 86°F), in a controlled room temperature environment. The medication should be kept out of reach of children and should not be used if the blister is torn or broken. The drug product in the package is from NDC 57237-018, Aising Health, LLC, and is distributed by American Health Packaging, Columbus, Ohio 43217.*
60 mg Duloxetine Delayed-Release Capsules Carton - 60 mg Carton

This is a description of a medication with the NDC code 60687-745-01, known as Duloxetine. It is a delayed-release capsule that contains 67.3mg of Duloxetine hydrochloride USP, equivalent to 80mg of Duloxetine. It comes in a package of 100 capsules (10 x 10) and is only available with a prescription. The pharmacist should dispense it with an accompanying medication guide for each patient's use. It is advised to keep it at a temperature of 20-25°C (68-77°F) and away from children. The package is distributed by American Health Packaging from Columbus, Ohio. The provided lot number is 57537-019, and the code has the following sequence 774501 047450111222.*
Figure 1: Cumulative Proportion of Adult Patients with MDD Relapse (Study MDD-5) - Figure1

The text is a graph showing the proportion of patients with relapse over time from randomization to relapse (in days) for two treatments: placebo and Duloxetine. The graph suggests that treatment with Duloxetine appears to have a lower proportion of patients experiencing relapse compared to placebo.*
Figure 10: Percentage of Adult Patients with OA Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity (Study OA-1) - Figure10

This appears to be a table showing the effectiveness of duloxetine, a medication used for pain management. The table displays the percentage of patients who experienced improvement in pain levels when taking doses of either 60 or 120 mg once daily, compared to a placebo. The x-axis shows the percentage of improvement in pain levels from baseline, while the y-axis shows the number of patients.*
Figure 2: Cumulative Proportion of Adult Patients with GAD Relapse (Study GAD-4) - Figure2

The text represents a graph or chart showing the proportion of patients with relapse over time from randomization to relapse in days. The chart shows two treatments: placebo and duloxetine, with duloxetine performing better as fewer patients relapse.*
Figure 3: Percentage of DPNP Adult Patients Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity (Study DPNP-1) - Figure3

Figure 4: Percentage of DPNP Adult Patients Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity (Study DPNP-2) - Figure4

Figure 5: Percentage of Adult Fibromyalgia Patients Achieving Various Levels of Pain Relief at Study Endpoint as Measured by 24-Hour Average Pain Severity (Study FM-1) - Figure5

This is a chart showing the percentage of patients who experienced improvement in pain from baseline using Duloxetine 60 mg Once Daily, Duloxetine 60 mg Twice Daily, and Placebo. The chart includes a range of percent improvement increments from 10 to 80.*
Figure 6: Percentage of Adult Fibromyalgia Patients Achieving Various Levels of Pain Relief at Study Endpoint as Measured by 24-Hour Average Pain Severity (Study FM-2) - Figure6
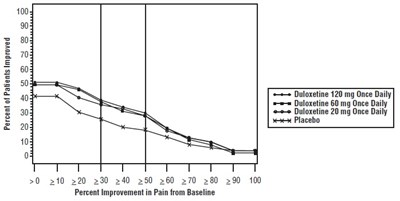
This is a graph showing the percentage of patients who experienced improvement in pain from baseline when taking Duloxetine at different dosages (20mg, 60mg, 120mg) once daily compared to those taking a placebo. The X-axis shows the percentage of improvement in pain and the Y-axis shows the percentage of patients.*
Figure 8: Percentage of Adult Patients with CLBP Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity (Study CLBP-1) - Figure8
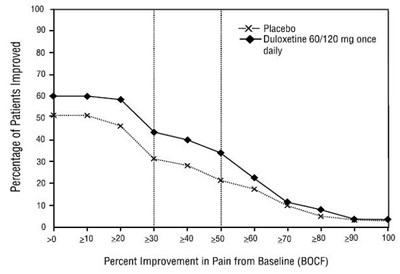
This document provides data on the percentage of patients who have experienced improvement after taking Duloxetine in comparison to the placebo. The data indicate that 100% of patients have improved, but it is unclear what specific symptoms or conditions are being measured. Additionally, the table presents information on percent improvement in pain from the baseline, but the values are not easily decipherable due to formatting issues.*
Figure 9: Percentage of Adult Patients with CLBP Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity (Study CLBP-3) - Figure9
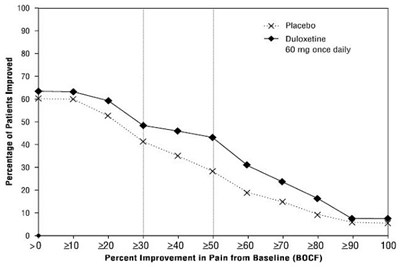
This document appears to be a chart or graph displaying the percentage of patients who improved while taking a placebo compared to those taking duloxetine at 60mg once daily. There is also a chart showing the percent improvement in pain from baseline. However, there is not enough information to determine what condition or study this data is referring to.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.





