Product Images Lurasidone Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Lurasidone Hydrochloride NDC 60687-769 by American Health Packaging, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
20 mg Lurasidone Hydrochloride Tablets Carton - 20 mg Carton

Lurasidone Hydrochloride Tablets under Axonly brand with NDC 60687-747-21. Each tablet contains 20 mg of Lurasidone Hydrochloride. The package contains 30 tablets (3 x 10) for pharmacy dispensation with the accompanying medication guide for patients. Usual Dosage should be followed as mentioned in the package insert for full prescribing information. Store the drug at a controlled room temperature of 20' 1025°C (68" to 77°F) excursions permitted between. The drug product should be kept out of reach of children and not be used if the blister is torn or broken. It is manufactured by Ascend Laboratories, LLC and distributed by American Health Packaging located in Columbus, Ohio.*
40 mg Lurasidone Hydrochloride Tablet Blister - 40 mg Blister
This is a listing of different tablets of Lurasidone Hydrochloride, each containing 40mg of the compound. The information includes details of the lot number, expiry date, and the type of tablet. It seems to be a collection of readings from different sources.*
40 mg Lurasidone Hydrochloride Tablets Carton - 40 mg Carton

This is a description of a medication package containing Lurasidone Hydrochloride Tablets. Each tablet has 40 mg of Lurasidone Hydrochloride (equivalent to 724 mg of Lurasidone). The package contains 30 tablets and should be stored between 20-25°C. The medication comes with a medication guide for the patient. The drug product in the package is from Ascend Laboratories, LLC. The package is distributed by American Health Packaging. Dosage information can be found in the package insert.*
60 mg Lurasidone Hydrochloride Tablet Blister - 60 mg Blister

This seems to be an output for a medication called "Lurasidone Hydrochloride Tablet" with a strength of 60 mg. However, due to the poor quality of the text and the inconsistent characters, it is not possible to determine the lot number and expiry dates.*
60 mg Lurasidone Hydrochloride Tablets Carton - 60 mg Carton

This is a description of a medication with the NDC code 60687-769-94. The medication is Lurasidone Hydrochloride and is in tablet form with a total of 20 tablets in each package. Each tablet contains 60mg of Lurasidone Hydrochloride, equivalent to 55.87 mg of Lurasidone. The usual dosage information can be found in the package insert. It is recommended to store the medication at a temperature between 20°C to 25°C (68°F to 77°F) with permissible excursions between 15°C to 30°C (53°F to 86°F). The medication should be kept out of reach of children and not used if the blister or packaging is torn or broken. The medication is distributed by American Health Packaging from Columbus, Ohio.*
Figure 2: Impact of Lurasidone Hydrochloride Tablets on Other Drugs - Figure2
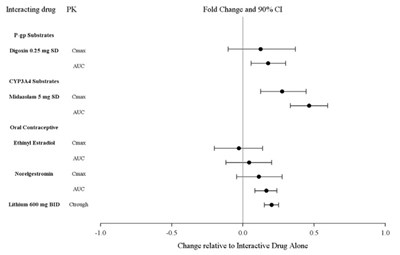
Not available. There is some medical jargon but without context, it is impossible to provide a useful description.*
Figure 3: Impact of Other Patient Factors on Lurasidone Hydrochloride Tablets Pharmacokinetics - Figure4
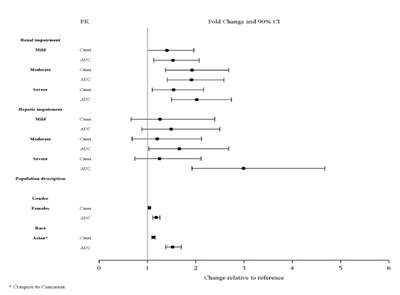
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.





