Product Images Topiramate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Topiramate NDC 60760-577 by St. Mary's Medical Park Pharmacy, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
top - Topiramate 60760 577 60

This is a description of NOG GHZ60-57 750 Topiramate medication, which comes in the form of tablets in 50mg. The package contains 60 tablets. The manufacturer is Cadia Healtheare Lid, located in Ahmedabad, India, and it was packaged by a company located in Oro Valley, AZ. The medication should be used as directed and requires a prescription from a managed pharmacy program. The medication should be stored at a controlled room temperature between 15°-30° C (59°-86°F).*
Structured product formula for Topiramate - a9eed983 edf1 4f44 a8ec 568a52f03015 02
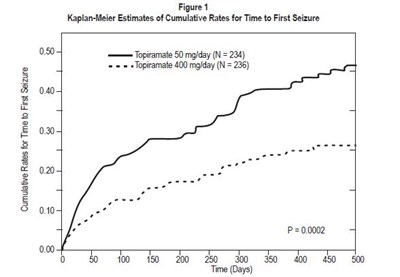
This is a graph representing the Cumulative Rates for Time to First Seizure using Kaplan-Meier Estimates. The text shows two groups of patients treated with different doses of Topiramate (50mg/day and 400mg/day) and their seizure rates over time (measured in days).*
Topiramate Tablets USP - a9eed983 edf1 4f44 a8ec 568a52f03015 03

This text presents a table showing the change from baseline to double-blind phase in the average monthly migraine period rate for different doses of topiramate and a placebo. The table includes the sample size (N) for each treatment group, but no other information is available to describe the study or the results.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
