Product Images Unithroid
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Unithroid NDC 60846-806 by Amneal Pharmaceuticals Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
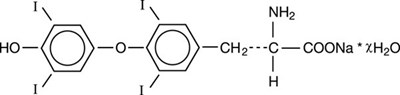
levothyroxine sodium tablets usp unithroid 11

This is a description of AdNITHROID sodium tablets, a medication used to treat hypothyroidism. It comes in a container that should be stored between 20-25°C (68-77°F), kept away from children, and dispensed in a tight, light-resistant container. The text includes the dosage of the tablets (175 meg or 0.175 mg) and mentions that it is Rx only. There is also a NDC code provided, which is 60846-810-01, and a revision date of 5/19. The rest of the text is not legible.*
levothyroxine sodium tablets usp unithroid 12

AdNITHROID® Sodium Tablets, USP is a prescription medication containing 200mcg (0.2mg) of Levothyroxine. It is dispensed in a tight, light-resistant container and should be kept out of reach of children. Storage conditions should not exceed 20-25°C (68-77°F) with excursions between 15-30°C (59-86°F). The product is supplied with 100 tablets and the National Drug Code (NDC) is 60846-811-01.*
levothyroxine sodium tablets usp unithroid 4

This is a prescription medication for 100 tablets of Levothyroxine Sodium Tablets, USP, with a dosage of 75mg. The medication should be kept out of reach of children and stored in a tight, light-resistant container at temperatures between 20-25°C (68-77°F). The medication is manufactured by a company with the NDC number 60846-803-01, and the label was last revised in May of 2019.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.