FDA Label for Wet Wipes
View Indications, Usage & Precautions
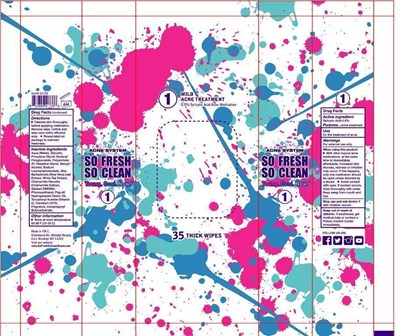
Wet Wipes Product Label
The following document was submitted to the FDA by the labeler of this product Hangzhou Guoguang Touring Commodity Co., Ltd.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient
salicylic acid 0.5%
Purpose
acne treatment
Use
for the treatment of acne
Warnings
for external use only
when using this product
with other topical acne medications ,at the same time or immediately afterwards ,increased skin dryness ,redness or
irritation may occur .If this happens ,only one medication should be used unless directed by a doctor.
avoid contact with eyes .if contact occurs, rinse thoroughly with water. keep away from mouth and lips.
stop use and ask doctor if skin irritation occurs.
Keep out of reach of children ,If swallowed ,get medical help or contact a Poison Control Center immediately.
Keep Out Of Reach Of Children
Keep out of reach of children ,If swallowed ,get medical help or contact a Poison Control Center immediately.
Directions
cleanse skin thoroughly before applying medication ,remove wipe ,unfold and wipe over entire affected area.
reseal label on package to maintain freshness.
Inactive Ingredient
aqua (water),glycerin, propylene glycol , dodecyl polyglucoside, polysorbate 20,HEXYLENE GLYCOL , BENZYL ALCOHOL ,SODIUM LAURAMINOPROPIONATE ,ALOE BARBADENSIS(ALOE VERA )EXTRACT ,WHITE TEA EXTRACT ,CHAMOMILLA RECUTITA(CHAMOMILE)EXTRACT ,GLYDANT DMDM H,PHENOXYETHANOL ,PEG-40 HYDROGENATED CASTOR OIL,TOCOPHERYL ACETATE(VITAMIN E) ,DISODIUM EDTA,IODOPROPYNYL BUTYLCARBAMATE
Package Label.Principal Display Panel
* Please review the disclaimer below.