Product Images Doxycycline
View Photos of Packaging, Labels & Appearance
- image description - Label
- image description - Molecule
- image description - Table1 1
- image description - Table1
- image description - Table2 1
- image description - Table2 2
- image description - Table2 3
- image description - Table2
- image description - Table3
- image description - Table4
- image description - Table5
- image description - Table6
- image description - Table7 1
- image description - Table7
Product Label Images
The following 14 images provide visual information about the product associated with Doxycycline NDC 61919-474 by Direct Rx, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image description - Label

The text provided contains details regarding the antibiotic medication Doxycycline, available in the form of tablets of 100 mg strength. The text further mentions the drug's generic variant - ADOXA and lists its main active ingredient as Doxycycline monohydrate. However, the text lacks clarity due to multiple errors and is difficult to understand fully.*
image description - Table1
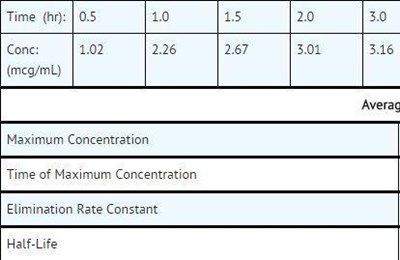
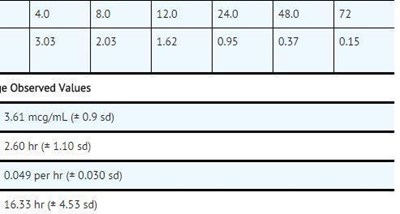
This is a data table showing different concentrations of a substance at different time points. The values are in units of meg/mL. The table also includes fields for calculating the average, maximum concentration, time of maximum concentration, elimination rate constant, and half-life. No further context or information is available.*
image description - Table2 1

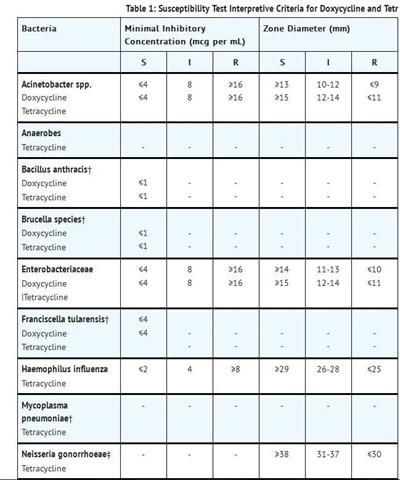
This appears to be a list of bacterial species and their susceptibility to the antibiotics doxycycline and tetracycline. The list includes Norcardiae, Actinomyces, Streptococcus pneumoniae, Vibrio cholera, Yersinia pestis, and Ureaplasma urealyticum. The effectiveness of the antibiotics varies for each species.*
image description - Table3

Doxycycline Tablets 50 mg available in blister packages of 10, cartons of 30 (3 x 10 unit dose tablets), bottles of 100, and bottles of 500. NDC codes provided for each package size.*
image description - Table4

Doxycycline Tablets 75 mg are available in blister packs of 10, cartons of 30 (5 x 10 unit dose tablets), bottles of 100, and bottles of 500. The NDC codes for each packaging size are listed.*
image description - Table5

Doxycycline Tablets 100 mg are available in various packaging options including blister packages of 10, cartons of 30 (3 x 10 unit dose tablets), bottles of 50, and bottles of 500. They are identified by NDC codes 23155-135-11, 23155-135-30, 23155-135-25, and 23155-135-05.*
image description - Table6
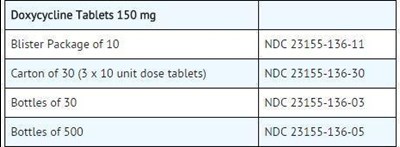
Doxycycline Tablets are available in various packaging including blister packages of 10, cartons of 30 (3 x 10 unit dose tablets), bottles of 30, and bottles of 500. The tablets have a strength of 150 mg each. The NDC codes for each package size are provided.*
image description - Table7 1
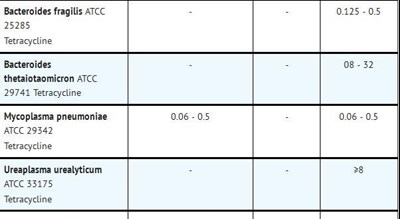
This text appears to be a list of four bacterial strains (Bacteroides fragilis ATCC 0125-05 25285, Bacteroides thetaiotaomicron ATCC 29741, Mycoplasma pneumoniae 006-05 ATCC 29342, and Ureaplasma urealyticum ATCC 33175) and the antibiotic Tetracycline.*
image description - Table7

This appears to be a table outlining the acceptable quality control ranges for susceptibility testing for the antibiotics Doxycycline and Tetracycline in several strains of bacteria, such as Enterococcus faecalis ATCC, Escherichia coli ATCC, Haemophilus influenzae ATCC, Neisseria gonorrhoeae ATCC, Staphylococcus aureus ATCC, and Streptococcus pneumoniae ATCC. The table features two methods of testing, minimal inhibitory zone diameter, and agar dilution concentration, and displays the acceptable ranges for each method.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.