Product Images Eye Itch Relief
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Eye Itch Relief NDC 62011-0232 by Strategic Sourcing Services Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
mck05 0005 02

This is an ophthalmic solution with antihistamine properties used only for the eyes, containing Ketotifen Fumarate. Distributed by McKesson from San Francisco, it is available under the brand name "HealthMart" with a money-back guarantee. The product is sterile and made in Switzerland. The bottle contains 5ml/ 0.17 fl oz and comes with directions on the carton. The storage temperature is between 20° to 25°C (68° to 77°F). The product has an NDC code of 62011-0232-1.*
mck05 0005 03
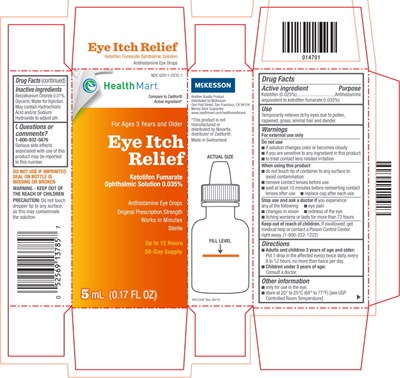
This is a drug information sheet for Ketoifen Fumarate Ophthalmic Solution, an antihistamine eye drop used for temporary relief of eye itchiness caused by pollen, ragweed, pets, etc. It provides directions for use, warnings, and precautions, including keeping the dropper from touching any surfaces, and reporting serious side effects to a certain number. The product is for external use only, and must not be used if the seal is missing or broken. The sheet also indicates that the product is not made or distributed by Novartis, the manufacturer of Zaditor® in Switzerland.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
