Product Images Dutasteride
View Photos of Packaging, Labels & Appearance
- Figure 1. AUA-SI Score* Change from Baseline (Randomized, Double-Blind, Placebo-Controlled Studies Pooled) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 02
- Figure 2. Percent of Subjects Developing Acute Urinary Retention Over a 24-Month Period (Randomized, Double-Blind, Placebo-Controlled Studies Pooled) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 03
- Figure 3. Percent of Subjects Having Surgery for Benign Prostatic Hyperplasia Over a 24-Month Period (Randomized, Double-Blind, Placebo-Controlled Studies Pooled) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 04
- Figure 4. Prostate Volume Percent Change from Baseline (Randomized, Double-Blind, Placebo-Controlled Studies Pooled) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 05
- Figure 5. Qmax Change from Baseline (Randomized, Double-Blind, Placebo-Controlled Studies Pooled) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 06
- Figure 6. International Prostate Symptom Score Change From Baseline Over a 48-Month Period (Randomized, Double-Blind, Parallel Group Trial [CombAT Trial]) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 07
- Figure 7. Qmax Change Ffrom Baseline Over a 24-Month Period (Randomized, Double-Blind, Parallel Group Study [CombAT Study]) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 08
- Dutasteride Capsules 0.5 mg bottle label (30 count) - dutasteride capsules bottle 30count
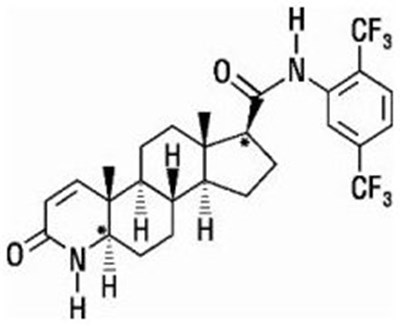
- Dutasteride structural formula - image 01
Product Label Images
The following 9 images provide visual information about the product associated with Dutasteride NDC 62112-8068 by Intergel Pharmaceuticals Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1. AUA-SI Score* Change from Baseline (Randomized, Double-Blind, Placebo-Controlled Studies Pooled) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 02

Figure 2. Percent of Subjects Developing Acute Urinary Retention Over a 24-Month Period (Randomized, Double-Blind, Placebo-Controlled Studies Pooled) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 03
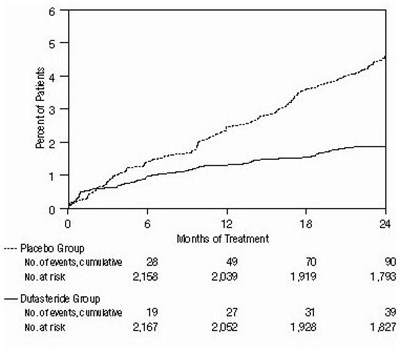
Figure 3. Percent of Subjects Having Surgery for Benign Prostatic Hyperplasia Over a 24-Month Period (Randomized, Double-Blind, Placebo-Controlled Studies Pooled) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 04
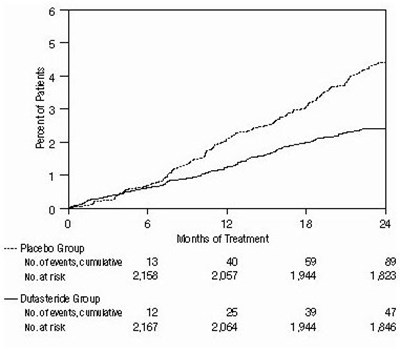
Figure 4. Prostate Volume Percent Change from Baseline (Randomized, Double-Blind, Placebo-Controlled Studies Pooled) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 05

Figure 5. Qmax Change from Baseline (Randomized, Double-Blind, Placebo-Controlled Studies Pooled) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 06

Figure 6. International Prostate Symptom Score Change From Baseline Over a 48-Month Period (Randomized, Double-Blind, Parallel Group Trial [CombAT Trial]) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 07
![7065c13c-25db-4b15-80d6-5fbb30dfeb0f-07.jpg Figure 6. International Prostate Symptom Score Change From Baseline Over a 48-Month Period (Randomized, Double-Blind, Parallel Group Trial [CombAT Trial]) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 07](https://ndclist.com/assets/spl/images/35f16489-00d7-0f8b-3cd6-2abbe277af19/400x-7065c13c-25db-4b15-80d6-5fbb30dfeb0f-07.jpg)
Figure 7. Qmax Change Ffrom Baseline Over a 24-Month Period (Randomized, Double-Blind, Parallel Group Study [CombAT Study]) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 08
![7065c13c-25db-4b15-80d6-5fbb30dfeb0f-08.jpg Figure 7. Qmax Change Ffrom Baseline Over a 24-Month Period (Randomized, Double-Blind, Parallel Group Study [CombAT Study]) - 7065c13c 25db 4b15 80d6 5fbb30dfeb0f 08](https://ndclist.com/assets/spl/images/35f16489-00d7-0f8b-3cd6-2abbe277af19/400x-7065c13c-25db-4b15-80d6-5fbb30dfeb0f-08.jpg)
Dutasteride Capsules 0.5 mg bottle label (30 count) - dutasteride capsules bottle 30count

This is a medication called Dutasteride with National Drug Code 62112-8068-1. Each capsule contains 0.5 mg of the drug. It is recommended to take one capsule per day without chewing or opening it. The prescription contains further dosage instructions. The capsules should be stored at Controlled Room Temperature between 15-30°C (59°-86°F). The medication should be dispensed in a sealed container, and usage is not recommended for women or children. There is also a warning to wash immediately with soap and water if there is contact with a leaking capsule.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.