Product Images Memantine Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 11 images provide visual information about the product associated with Memantine Hydrochloride NDC 62332-075 by Alembic Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - memantine fig1
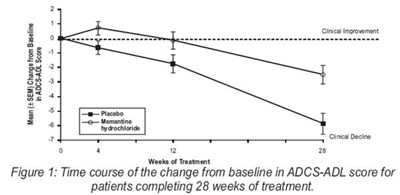
The text describes a figure labeled as "Figure 1" that shows the time course of the change from baseline in ADCS-ADL score for patients who completed 28 weeks of treatment with Memantine hydrochloride. The figure presents a mean score (+SEM) range from baseline and reports clinical improvement and decline.*
Figure 2 - memantine fig2
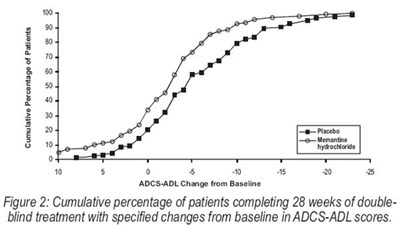
The text describes a figure labeled as "Figure 2", which shows the cumulative percentage of patients who completed 28 weeks of double-blind treatment and their changes from baseline in ADCS-ADL scores. The figure has a vertical axis showing percentages and a horizontal axis showing score changes from baseline (ranging from -10 to 30). The text does not provide further context or information.*
Figure 4 - memantine fig4
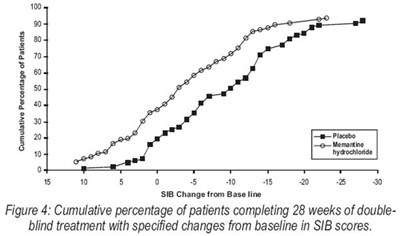
This is a graph, named "Figure 4", that displays the percentage of patients who completed a 28-week double-blind treatment and the changes from baseline in SIB (Severe Impairment Battery) scores. The treatment is related to placebo and memantine hydrochloride.*
Figure 8 - memantine fig8
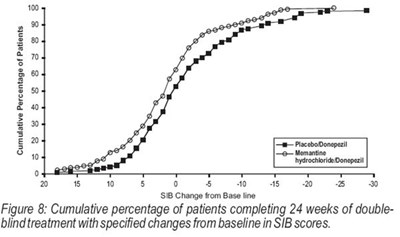
This is a figure showing the cumulative percentage of patients who completed 24 weeks of double-blind treatment with specified changes from baseline in SIB scores, using Donepezil and Memantine hydrochloride.*
Figure 3 - memantine figure3
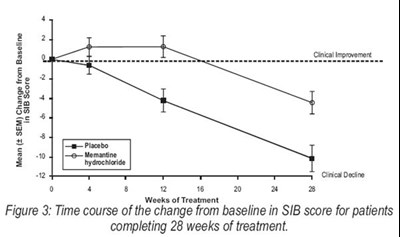
The text describes a figure displaying the time course of change in SIB score for patients who completed 28 weeks of treatment. The figure shows the mean change from baseline using the drug memantine hydrochloride. There is no additional information available in the given text.*
Figure 5 - memantine figure5
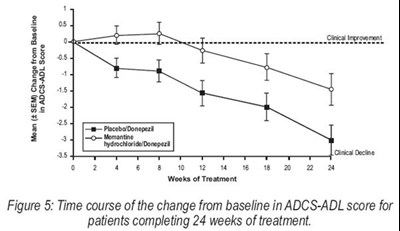
The text describes a figure labeled as "Figure 5" that presents a graph showing the time course of change from baseline in ADCS-ADL score for patients who underwent 24 weeks of treatment. The mean and SEM are also provided in the figure. The text does not provide any further context or details, making it difficult to understand the purpose or results of the study.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




