Product Images Arformoterol Tartrate
View Photos of Packaging, Labels & Appearance
- arformoterol-1s-30-carton - arformoterol 1s 30 carton
- arformoterol-1s-foil - arformoterol 1s foil
- arformoterol 4s 15 carton
- arformoterol-4s-foil - arformoterol 4s foil
- arformoterol-fig1 - arformoterol fig1
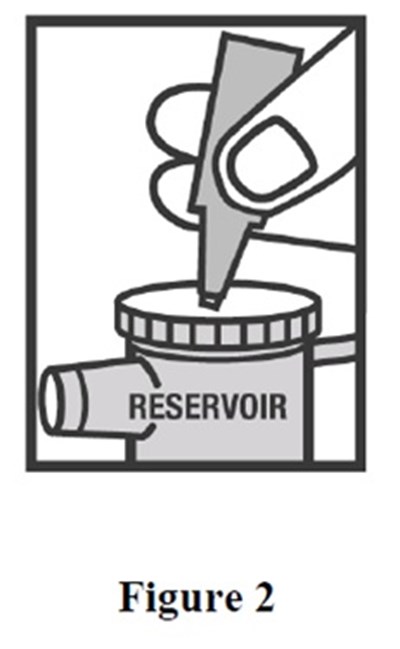
- arformoterol-fig2 - arformoterol fig2
- arformoterol-fig3-4 - arformoterol fig3 4
- arformoterol-fig5 - arformoterol fig5
- arformoterol-fig6 - arformoterol fig6
- arformoterol-graph1-new.jpg - arformoterol graph1 new
- arformoterol-graph2-new.jpg - arformoterol graph2 new
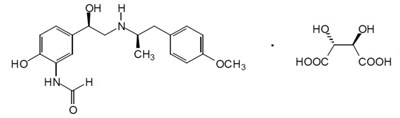
- arformoterol-structure - arformoterol structure
Product Label Images
The following 12 images provide visual information about the product associated with Arformoterol Tartrate NDC 62332-633 by Alembic Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
arformoterol-1s-foil - arformoterol 1s foil

Arformoterol Tartrate Inhalation Solution is a medication that comes in a pre-measured dose of 15 micrograms per 2 milliliters. It is packaged in a pouched form and can be stored at room temperature for up to 2 weeks before expiring. The medication should not be used after the expiration date or discarded improperly. The drug is intended for inhalation use only and should not be administered in any other way.*
arformoterol 4s 15 carton

Arformoterol Tartrate Inhalation Solution is a prescription drug that is available in a 15 meg/2 mL dosage, and the potency expressed is as arformoterol. It should be stored in a refrigerator or at room temperature for up to two weeks. This medication is for oral use only and is meant for patients with breathing complications. The text also includes the National Drug Code and dosage information for the prescription.*
arformoterol-4s-foil - arformoterol 4s foil

Arformoterol Tartrate is an inhalation solution. Each single unit dose vial contains 15 mig of aromoterol, which is equivalent to 0.2 mg of arformoterol tartrate in a sterile, isotonic saline solution with 1.5% w/v of benzalkonium chloride and sodium chloride. After opening the pouch, the unused vials should be kept out of the reach of children and those exposed to excessive heat. Unopened pouched unit dose vials can be stored in a refrigerator (2°C to 8°C, 36°F to 46°F). It's recommended to use only as directed by a physician and do not exceed the recommended dosage. The manufacturer is Nombic Pharmaceuticals.*
arformoterol-fig3-4 - arformoterol fig3 4
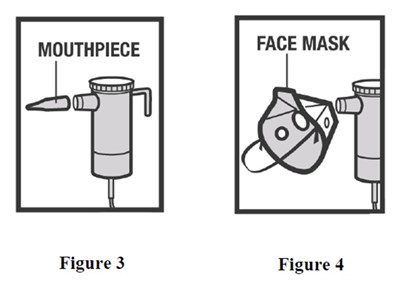
This is a product description for a face mask. There are two figures included, but it is not possible to determine what they illustrate without further context.*
arformoterol-graph1-new.jpg - arformoterol graph1 new
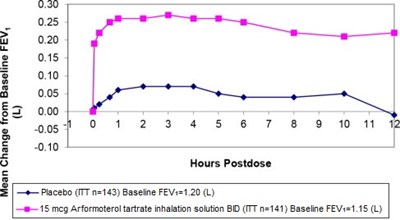
This appears to be a graph showing changes in FEV1 (a measure of lung function) over time (up to 12 hours post-dose) in two groups of patients: one group receiving a placebo and another group receiving 15 micrograms of arformoterol tartrate inhalation solution twice daily. The baseline FEV1 measurements for the two groups were 120 and 1.15 liters, respectively.*
arformoterol-graph2-new.jpg - arformoterol graph2 new
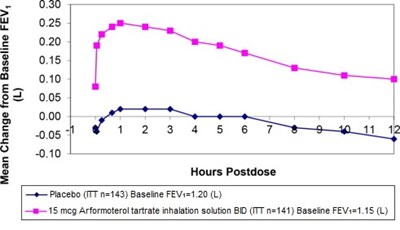
This appears to be a graph showing the change from baseline forced expiratory volume (FEV) over time for a placebo group and a group receiving a 15 meg Arformoterol tartrate inhalation solution twice daily. The x-axis represents hours postdose and the y-axis represents the change in FEV from baseline.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.