Product Images Formoterol Fumarate
View Photos of Packaging, Labels & Appearance
- 30s pack label
- 60s pack label
- formoterol-figure-1 - formoterol figure 1
- formoterol-figure-2 - formoterol figure 2
- formoterol-instruction-image-1 - formoterol instruction image 1
- formoterol-instruction-image-2 - formoterol instruction image 2
- formoterol-instruction-image-3 - formoterol instruction image 3
- formoterol-structure - formoterol structure
- pouch label
Product Label Images
The following 9 images provide visual information about the product associated with Formoterol Fumarate NDC 62332-655 by Alembic Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
60s pack label

This text provides information about the storage conditions and usage instructions of Formoterol Fumarate Inhalation Solution, which is contained in 2 mL vials. The product should be stored refrigerated prior to dispensing to the patient, and after dispensing, it should be stored in a refrigerator or at room temperature and discarded when the drug expires or not used after 3 months. The pouch containing the vial should be protected from light and heat. The text also contains information about the manufacturer, the inactive and active ingredients, and the dosage.*
formoterol-figure-1 - formoterol figure 1
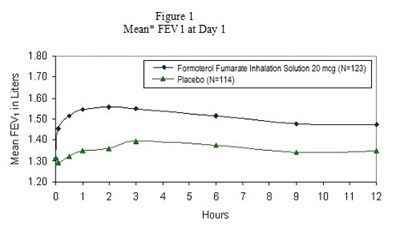
The text describes a graph or chart called "Mean FEVy in Liters" with data shown for two groups: "Fomorol Fumara rhaason Sokson 20g (N-123)" and "Piacebo (N=114)". The graph also includes a label for "Mean FEV 12t Day 1".*
formoterol-figure-2 - formoterol figure 2

The text describes a graph (Figure 2) that shows the mean FEV1 (Forced Expiratory Volume in 1 second) at the endpoint after 12 weeks of treatment with Formoterol Fumarate Inhalation Powder 20g in 123 patients. The graph also shows the mean FEVa (most likely Forced Expiratory Volume in Liters).*
pouch label

This is a pharmaceutical product description for an inhalation drug. It is a 20 meg/2 mL vial for oral inhalation only. The active ingredient is Formoterol fumarate, USP, while the inactive ingredients are Citric acid, Sodium Citrate, Sodium Chloride, and Water for injection. The drug should be stored in a refrigerator at 2°C to 8°C or room temperature, 20°C to 25°C, and discarded when expired or unused for three months. It should be protected from light and heat and kept out of reach of children. The description warns users to read the accompanying patient information and the drug is available only by prescription.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




