Product Images Amlodipine And Atorvastatin
View Photos of Packaging, Labels & Appearance
- amloatorva-10-10 - amloatorva 10 10
- amloatorva-10-20 - amloatorva 10 20
- amloatorva-10-40 - amloatorva 10 40
- amloatorva-10-80 - amloatorva 10 80
- amloatorva-25-10 - amloatorva 25 10
- amloatorva-25-20 - amloatorva 25 20
- amloatorva-25-40 - amloatorva 25 40
- amloatorva-5-10 - amloatorva 5 10
- amloatorva-5-20 - amloatorva 5 20
- amloatorva-5-40 - amloatorva 5 40
- amloatorva-5-80 - amloatorva 5 80
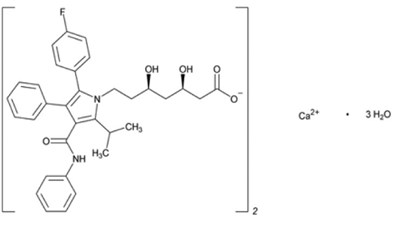
- amloatorva-amlodipine.jpg - amloatorva amlodipine
- amloatorva-atorvastatin.jpg - amloatorva atorvastatin
- Figure 1 - amloatorva fig 1
- amloatorva-fig3.jpg - amloatorva fig3
- amloatorva-fig4.jpg - amloatorva fig4
- amloatorva-fig5.jpg - amloatorva fig5
- amloatorva-figure-2.jpg - amloatorva figure 2
Product Label Images
The following 18 images provide visual information about the product associated with Amlodipine And Atorvastatin NDC 62332-759 by Alembic Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
amloatorva-10-10 - amloatorva 10 10

This is a description of a medication containing 10 mg of amlodipine (equivalent to 13.9 mg amlodipine besylate) and 10mg of atorvastatin (equivalent to 10.3 mg atorvastatin calcium). The product is in tablet form and has specific dosage and usage instructions to be followed as per the prescribing information provided. The medication is manufactured by Alembic Pharmaceuticals Limited in India for Alembic Pharmaceuticals, Inc. in the USA. The tablet is not to be sold without a prescription (Rx only).*
amloatorva-10-20 - amloatorva 10 20

This text contains information about a medication that is a combination of amlodipine and atorvastatin. It specifies the dosage and manufacturer details. The tablets are intended for lowering blood pressure and cholesterol levels. The packaging is child-resistant and the storage conditions are indicated. It also provides a GTIN code and manufacturing license number.*
amloatorva-10-40 - amloatorva 10 40

This text provides information about a medication that contains 10 mg of amlodipine. It also indicates the manufacturer as Alembic Pharmaceuticals and the dosage details on the accompanying tablets. The medication appears to be a combination of amlodipine and atorvastatin. The prescription is for 30 tablets and is for use in the USA.*
amloatorva-10-80 - amloatorva 10 80

This text appears to be a description of a medication packaging for tablets containing a combination of amlodipine besylate and atorvastatin. The tablets contain 10mg of amlodipine (equivalent to 13.9mg amlodipine besylate) and 80mg of amlodipine and atorvastatin (equivalent to 82.7mg atorvastatin calcium). The dosage and use information can be found in the accompanying prescribing information. The tablets are manufactured by Alembic Pharmaceuticals Limited in Gujarat, India, and are distributed in the USA by Alembic Pharmaceuticals, Inc.*
amloatorva-25-10 - amloatorva 25 10

This text provides information about a medication containing a combination of amlodipine and atorvastatin, with each tablet containing specific amounts of the active ingredients. It also includes details about the manufacturer, dosage, and use. The medication is in tablet form and is indicated for prescription use only. This package contains 30 tablets and has specific storage instructions. It is manufactured by Alembic Pharmaceuticals Limited for distribution in the USA.*
amloatorva-25-20 - amloatorva 25 20

This text contains information about a medication tablet that includes a combination of amlodipine (equivalent to 3.5 mg) and aliskiren (equivalent to 20 mg). It also mentions other ingredients such as valsartan and atorvastatin calcium. The dosage and use instructions can be found in the accompanying prescribing information. The tablets are manufactured by Alembic Pharmaceuticals Limited in India and are intended for prescription use. Additionally, the text mentions a pharmaceutical company located in Bedminster, NJ, USA.*
amloatorva-25-40 - amloatorva 25 40

This text provides information about a medication that contains a combination of amlodipine and atorvastatin. Each tablet contains 2.5 mg of amlodipine and 41.4 mg of atorvastatin. The dosage and use instructions should be referred to the accompanying prescribing information. The medication is manufactured by Alembic Pharmaceuticals Limited in India and distributed by Alembic Pharmaceuticals, Inc. in the USA. The package contains 30 tablets. The expiration date is listed as December 2024, and proper storage recommendations are provided including a controlled room temperature range of 15° to 30°C (59° to 86°F). The packaging is child-resistant.*
amloatorva-5-10 - amloatorva 5 10

This is the fine print containing information about a medication. The tablet contains 5 mg of amlodipine (equivalent to 6.9 mg of amlodipine besylate, USP) and 10 mg of atorvastatin calcium, USP. It provides details on dosage, use, and prescribing information. The medication is manufactured by Alembic Pharmaceuticals Limited in India and is for prescription use only. The product is distributed by Alembic Pharmaceuticals, Inc. in Bedminster, NJ, USA.*
amloatorva-5-20 - amloatorva 5 20

This is a medication containing a combination of amlodipine (calcium channel blocker) and atorvastatin (statin) to treat high blood pressure and high cholesterol levels. Each tablet contains 5mg of amlodipine and 20 mg of atorvastatin. The dosage and usage information can be found in the accompanying prescribing information. Manufactured by Alembic Pharmaceuticals Limited in India and marketed by Alembic Pharmaceuticals, Inc. in the USA. The package is child-resistant.*
amloatorva-5-40 - amloatorva 5 40

This product is a combination tablet containing 5mg of amlodipine (equivalent to 6.9 mg amlodipine besylate) and 40 mg of atorvastatin (equivalent to 41.4 mg atorvastatin calcium). It is manufactured by Alembic Pharmaceuticals Limited in India and distributed in the USA by Alembic Pharmaceuticals, Inc. The tablets are indicated for prescription use only with dosage information provided in the prescribing information. The product is labeled as Amlodipine and Atorvastatin Tablets, USP, with a total of 30 tablets in a package. The storage temperature is recommended to be between 15°C to 30°C (59°F to 86°F), with the package being child-resistant. Additional information includes an NDC code for identification and manufacturing license details.*
amloatorva-5-80 - amloatorva 5 80

This text appears to be a detailed information about a medication in tablet form. It includes the content of the tablets which contain a combination of amlodipine and atorvastatin in specified amounts. The dosage and use instructions are likely present in this text, along with details such as manufacturer information and the number of tablets per package. It also mentions that it is a prescription-only medication.*
Figure 1 - amloatorva fig 1

This text seems to be a data table showing event rates over time for a placebo and Amlodipine. The table includes information on the number of events at different time points in months. The data presented could be useful for evaluating the effectiveness of the Amlodipine treatment compared to the placebo in reducing certain events over time.*
amloatorva-fig3.jpg - amloatorva fig3

This text provides a comparison between Atorvastatin and Placebo in a study, showing HR=0.64 (0.5 to 0.83) with a p-value of 0.0005 over a timeline of 0 to 3.5 years. It indicates a potential positive impact or benefit associated with the use of Atorvastatin over Placebo in the context of the study.*
amloatorva-fig4.jpg - amloatorva fig4

This is information related to a clinical study comparing the cumulative hazard (%) between a placebo group and a group using Atorvastatin over a four-year follow-up period. The Hazard Ratio (HR) was calculated to be 0.63 with a 95% confidence interval of 0.48 to 0.83, indicating a significant difference between the two groups. The p-value was found to be 0.001. The data suggests that Atorvastatin may be associated with a lower risk of the primary endpoint compared to the placebo.*
amloatorva-fig5.jpg - amloatorva fig5

This data appears to show the percentage of subjects experiencing an event under different treatment regimens of atorvastatin (10 mg and 80 mg). It cites a hazard ratio (HR) of 0.78 with a confidence interval and a p-value. The chart may be depicting the time to the first major cardiovascular endpoint over a span of years for the mentioned treatments.*
amloatorva-figure-2.jpg - amloatorva figure 2

This text provides information on a study comparing the effects of Amlodipine and Placebo on a group of patients. The data is segmented based on age, gender, baseline SBP, presence of vessel stenosis, and PCI status. The hazard ratio with a 95% confidence interval is also mentioned. The study mentions that the mean baseline SBP of the patients is 120 mmHg.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.