Product Images Ondansetron Hydrochloride
View Photos of Packaging, Labels & Appearance
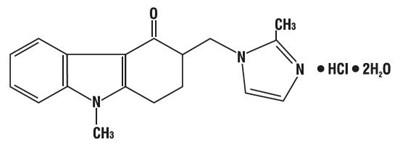
- chemical-structure - chemical structure
- showbox4mg - showbox4mg
- showbox8mg - showbox8mg
- spl-ondansetron-chemical-structure - spl ondansetron chemical structure
- spl-ondansetron-hcl-label-4mg - spl ondansetron hcl label 4mg
- spl-ondansetron-hcl-label-8mg - spl ondansetron hcl label 8mg
- spl-ondansetron-odt-4mg-showbox - spl ondansetron odt 4mg showbox
- spl-ondansetron-odt-8mg-showbox - spl ondansetron odt 8mg showbox
- spl-ondansetron-odt-label-4mg - spl ondansetron odt label 4mg
- spl-ondansetron-odt-label-8mg - spl ondansetron odt label 8mg
Product Label Images
The following 10 images provide visual information about the product associated with Ondansetron Hydrochloride NDC 62756-131 by Sun Pharmaceutical Industries, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
showbox4mg - showbox4mg

This is a medication information for Ondansetron Hydrochloride Tablets, with National Drug Code (NDC) number 62756-130-02. The tablets are available in 4mg dosage and require a prescription (Rx only). The tablets are packaged in a box containing 10 blister packs, each pack with 10 unit dose tablets for a total of 100 tablets.*
showbox8mg - showbox8mg

This is a prescription drug labeled as Ondansetron Hydrochloride Tablets with a strength of 8mg. It is available in 100 unit dose tablets which come in packs of 10x10. The NDC or National Drug Code for the drug is 62756-131-02.*
spl-ondansetron-hcl-label-4mg - spl ondansetron hcl label 4mg

Ondansetron Hydrochloride Tablets are available in film-coated form and are prescribed for oral consumption. Each tablet has 5 mg of ondansetron hydrochloride, USP (dihydrate) which is equal to 4 mg of ondansetron. The packaging insert provides details for dose and administration. The Tablets should be stored at controlled room temperature 20° to 25°C (68° to 77°F) and kept away from light. Dispensing should be done in a tight, light-resistant container as defined by the USP. These tablets are manufactured by Sun Pharmaceutical Industries Ltd. Halol-Baroda Highway, Halol-389 350, Gujarat, India, and distributed by Sun Pharmaceuticals Ltd. Cranbury, NJ 08512. The Product code is NDC 62756-130-01.*
spl-ondansetron-hcl-label-8mg - spl ondansetron hcl label 8mg

Each film-coated tablet of Ondansetron manufactured by Sun Pharmaceutical Industries Ltd contains 10 mg of ondansetron hydrochloride. The usual dosage to be seen in the package insert for dosage and administration. It is recommended to store it at a temperature of 20°C to 25°C, protect from light and dispense in a tight, light-resistant container as defined in the USP. This medication is only available with a prescription and is distributed by Sun Pharmaceutical Industries, Inc. in Cranbury, NJ.*
spl-ondansetron-odt-4mg-showbox - spl ondansetron odt 4mg showbox

This is a medication called Ondansetron, which comes in the form of orally disintegrating tablets. The package contains 30 tablets in total, split into 3 blister cards each with 10 tablets. The indicated use for this medicine is not mentioned, but it is only available through a prescription (Rx only). The NDC (National Drug Code) number is 62756-240-64, and the drug is manufactured by SIJN PHARMA.*
spl-ondansetron-odt-8mg-showbox - spl ondansetron odt 8mg showbox

This is a prescription medicine called Ondansetron which comes in a pack of 30 orally disintegrating tablets in three blister cards with ten tablets each. The manufacturer is Sun Pharma. NDC number 62756-356-64 is also mentioned.*
spl-ondansetron-odt-label-4mg - spl ondansetron odt label 4mg

This is a description of ondansetron orally disintegrating tablets. Each tablet contains 4mg of ondansetron and includes a warning to phenylketonurics due to the presence of phenylalanine. Dosage and administration information can be found in the package insert. The tablets must be stored at temperatures between 20°C to 25°C (68°F to 77°F). The product disintegrates within approximately 60 seconds, although it does not meet the USP Disintegration Time. The tablets are distributed by Sun Pharmaceutical Industries, Inc. located in Cranbury, NJ and the packaging contains 30 tablets. The manufacturer, Sun Pharmaceutical Industries Ltd., is located in Halol-Baroda Highway, Halol-389 350, Gujarat, India.*
spl-ondansetron-odt-label-8mg - spl ondansetron odt label 8mg

Each tablet of this medication contains 8mg of ondansetron, USP. The medication may contain phenylalanine, and patients with phenylketonuria should be cautious. Dosage and storage information can be found in the package insert. The medication disintegrates in approximately 60 seconds but it does not meet USP disintegration time. It is available in a 30 tablet package and requires a prescription. This product is manufactured by Sun Pharmaceutical Industries Ltd. in Gujarat, India and distributed by Sun Pharmaceutical Industries Inc. in Cranbury, NJ.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.