Product Images Sezaby
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Sezaby NDC 62756-301 by Sun Pharmaceutical Industries, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image description - sezaby 2
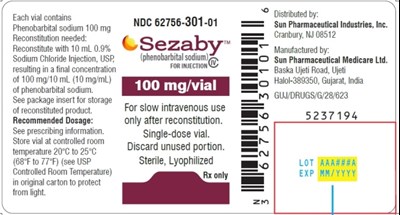
This is a pharmaceutical product containing Phenobarbital sodium, with each vial containing 100 mg. It requires reconstitution with 10 mL 0.9% Sodium Chloride Injection, USP, resulting in a final concentration of 100 mg/10 mL (10 mg/ml) of Phenobarbital sodium. It is for slow intravenous use only after reconstitution, and is a single-dose vial. The recommended dosage and storage information can be found in the package insert. It is distributed by Sun Pharmaceutical Industries, Inc. and manufactured by Sun Pharmaceutical Medicare Ltd. in India. The lot and expiration date are also provided.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

