Product Images Fexofenadine Hydrochloride
View Photos of Packaging, Labels & Appearance
- 180mg-allergy-1 - 180mg allergy 1
- 180mg-allergy-2 - 180mg allergy 2
- 180mg-allergy-sb - 180mg allergy sb
- 30mg-allergy-2 - 30mg allergy 2
- 30mg-allergy-3 - 30mg allergy 3
- 30mg-allergy-4 - 30mg allergy 4
- 30mg-allergy-5 - 30mg allergy 5
- 30mg-allergy-6 - 30mg allergy 6
- 30mg-allergy-7 - 30mg allergy 7
- 30mg-allergy-sb - 30mg allergy sb
- 60mg-allergy-1 - 60mg allergy 1
- 60mg-allergy-2 - 60mg allergy 2
- 60mg-allergy-3 - 60mg allergy 3
- 60mg-allergy-sb - 60mg allergy sb
Product Label Images
The following 14 images provide visual information about the product associated with Fexofenadine Hydrochloride NDC 62756-543 by Sun Pharmaceutical Industries, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
180mg-allergy-1 - 180mg allergy 1

This is a description for a box of Fexofenadine Hydrochloride tablets to relieve allergies. It warns that the tablets are non-drowsy and not to use them if the inner safety seal is missing or torn. The box contains 100 tablets of 180mg each and is suitable for the relief of indoor and outdoor allergies. There are also instructions for use on the label. Finally, there are various symbols and codes at the bottom of the box, but it is not clear what they represent.*
180mg-allergy-sb - 180mg allergy sb

This is the drug facts section for Fexofenadine Hydrochloride tablets. It includes information on the inactive ingredients, the National Drug Code, dosage, and potential warnings. The medication is an antihistamine used for allergy relief both indoors and outdoors, with specific benefits for itchy nose, throat, and eyes. The product comes in blister packaging and should not be used if the package is damaged. Directions indicate dosage and age restrictions. The remainder of the text is unclear due to poor quality.*
30mg-allergy-2 - 30mg allergy 2
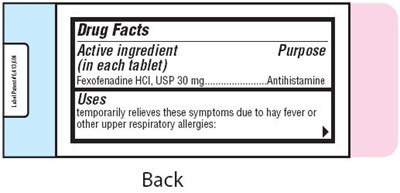
Drug Facts table with information on the active ingredient Fexofenadine HCI, which is an antihistamine commonly used to temporarily relieve symptoms caused by hay fever or other upper respiratory allergies. Unfortunately, there is no additional information available to provide a more detailed description.*
30mg-allergy-3 - 30mg allergy 3

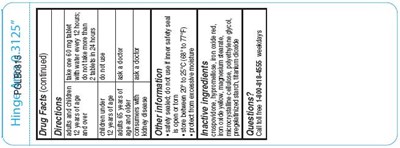
This is a drug label that provides instructions and warnings for a product that may be used to treat symptoms such as a runny nose, sneezing, itchy and watery eyes, and itchiness of the nose or throat. It advises users to consult a doctor if they have kidney disease and to not exceed the recommended dosage. It also warns against taking the product with certain substances and for individuals who have had an allergic reaction to the product or its ingredients. The label includes inserts for the top and back of the product.*
30mg-allergy-4 - 30mg allergy 4

This text is a continuation of the drug facts section of a product. It includes instructions on when to stop using the product and seek medical help, precautions for pregnant or breastfeeding individuals, and directions for use. The product is in tablet form, with different dosages for adults and children over and under 12 years old. The text also includes an instruction to keep the product out of reach of children and what to do in case of an overdose. There are also two inserts mentioned but not described.*
30mg-allergy-5 - 30mg allergy 5

This is a description of a drug's additional information. The text warns against the use of the drug by children below the age of 5, adults above the age of 65 and people with kidney disease. It advises to consult a doctor before taking the drug. The drug comes safety sealed, and one should not use it if the safety seal is broken. It should be stored at a temperature between 20° to 25°C (66° to 77°F) and protected from moisture. The drug contains ypromeIlose and iron oxide.*
30mg-allergy-6 - 30mg allergy 6

This is a continuation of drug facts information. The text mentions several ingredients used in the drug such as iron oxide yellow, magnesium stearate, microcrystalline cellulose, polyethylene glycol, pregelatinized starch, and titanium dioxide. A toll-free number is provided for any questions. The last sentence mentions an "Insert 4Top" and "Insert 4 Back," but it's not clear what it refers to.*
30mg-allergy-7 - 30mg allergy 7

Children's Fexofenadine Hydrochloride Tablets, USP is an antihistamine used for indoor and outdoor allergies. It's non-drowsy and suitable for ages 6 years and older. The medicine comes in a pack of 100 tablets, each containing 30mg of the active ingredient. The manufacturer is Sun Pharmaceutical Industries Ltd. The label advises not to use if the inner safety seal is torn or missing and carries the warning "SEALED FOR YOUR PROTECTION."*
30mg-allergy-sb - 30mg allergy sb

Product Name: Children's Fexofenadine Hydrochloride Tablets, USP, RGN prescription strength. This text also includes some illegible characters and lines of incomplete or unclear text.*
60mg-allergy-sb - 60mg allergy sb

This is a drug information label for Fexofenadine Hydrochloride Tablets, USP. The tablets contain 60mg of the active ingredient and are prescribed to relieve allergy symptoms.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.