Product Images Doxorubicin Hydrochloride
View Photos of Packaging, Labels & Appearance
- doxorubicin-carton-200mg - doxorubicin carton 200mg
- doxorubicin-carton-50mg - doxorubicin carton 50mg
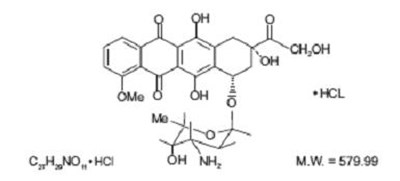
- doxorubicin-chem-str - doxorubicin chem str
- doxorubicin-fig01 - doxorubicin fig01
- doxorubicin-fig02 - doxorubicin fig02
- doxorubicin-label-200mg - doxorubicin label 200mg
- doxorubicin-label-50mg - doxorubicin label 50mg
Product Label Images
The following 7 images provide visual information about the product associated with Doxorubicin Hydrochloride NDC 62756-827 by Sun Pharmaceutical Industries, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
doxorubicin-carton-200mg - doxorubicin carton 200mg
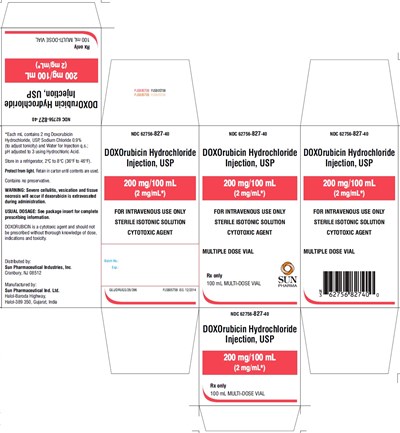
This is a medication called DOXOrubicin Hydrochloride, used for intravenous administration. Each mL of the solution contains 2mg of Doxorubicin Hydrochloride and is isotonic with sodium chloride 0.9%. The pH of the solution has been adjusted using Hydrochloric Acid. The medication should be stored under refrigeration at 2°C to 8°C (36°F to 46°F) and protected from light. This is a cytotoxic agent and severe cellulitis, vesication and tissue necrosis can occur if it is extravasated during administration. Please refer to the package insert for complete prescribing information. The medication is distributed by Sun Pharmaceutical Industries, Inc. and manufactured by Sun Pharmaceutical Ind. Ltd. The medication is presented in a multi-dose vial of 100mL. *
doxorubicin-carton-50mg - doxorubicin carton 50mg

This is the label for DOXOrubicin Hydrochloride Injection, a cytotoxic agent used intravenously to treat cancer. It contains 2 mg Doxorubicin Hydrochloride, USP Sodium Chioide 0.9% and Water for Infections. It is important to store it between 2-8 degrees Celsius, keep it out of light, and use caution during administration to prevent extravasation. Detailed prescribing information is included in the package. The drug is manufactured in India and distributed by Sun Pharmaceutical Industries, Inc.*
doxorubicin-fig01 - doxorubicin fig01
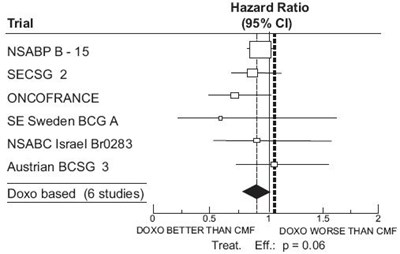
The text is a table showing different trials and their corresponding hazard ratios with their confidence intervals. A hazard ratio is a measure of the effect of a treatment or intervention on an outcome, such as survival, in a clinical trial. The text shows several clinical trials and their associated hazard ratios.*
doxorubicin-label-200mg - doxorubicin label 200mg

Each mL of this injection contains 2mg of Doxorubicin Hydrochloride, USP Sodium Chloride 0.9% and Water for Infection. No preservatives are included. The usual dosage information can be found in the package insert. This is a cytotoxic agent and severe cell damage, vesication, and tissue necrosis may occur if Doxorubicin is extravasated during administration. Store in a refrigerator at 2°C to 8°C (36°F to 46°F) and protect from light. This is a multiple dose vial for intravenous use only. The manufacturer is Sun Pharmaceutical Industries, and the injection has a National Drug Code (NOC) of 62756-827-40.*
doxorubicin-label-50mg - doxorubicin label 50mg

This is a description of DOXOrubicin Hydrochloride Injection, USP used for intravenous use. Each vial contains 50 mg of doxorubicin hydrochloride, USP in 25 mL of a sterile isotonic solution. The pH level is adjusted to 3 using hydrochloric acid. It is recommended to store the vial in a refrigerator at 2°C to 6°C and protect it from light until the time of use. Severe cellulitis, desiccation, and tissue necrosis may occur if the medication is extravasated during administration. The usual dosage details can be found in the package insert. The text also includes information about the distributor and the manufacturer of the medication.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.