Product Images Lenvima
View Photos of Packaging, Labels & Appearance
- The chemical structure of lenvatinib mesylate is LENVIMA, a kinase inhibitor, is the mesylate salt of lenvatinib. Its chemical name is 4-[3-chloro-4-(N’-cyclopropylureido)phenoxy]-7-methoxyquinoline-6 carboxamide methanesulfonate. The molecular formula is C21H19ClN4O4 • CH4O3S, and the molecular weight of the mesylate salt is 522.96. - lenvima 01
- Figure 1: Kaplan-Meier Curves for Progression-Free Survival in - lenvima 02
- Figure 2: Kaplan-Meier Curves for Progression-Free Survival in Study 205 - lenvima 03
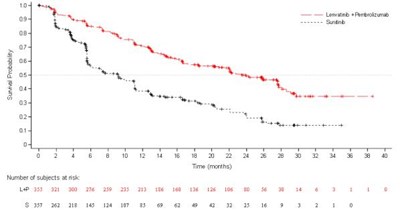
- Figure 3: Kaplan-Meier Curves for Overall Survival in Study 205 - lenvima 04
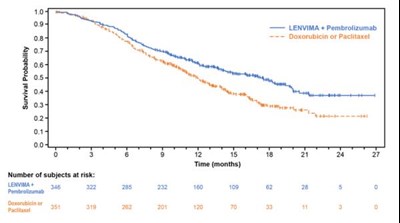
- Figure 4: Kaplan-Meier Curves for Overall Survival in REFLECT - lenvima 05
- PRINCIPAL DISPLAY PANELNDC 62856-704-30Lenvima(lenvatinib) capsules4 mg daily dose - lenvima 06
- PRINCIPAL DISPLAY PANELNDC 62856-708-05Lenvima(lenvatinib) capsules8 mg daily dose - lenvima 07
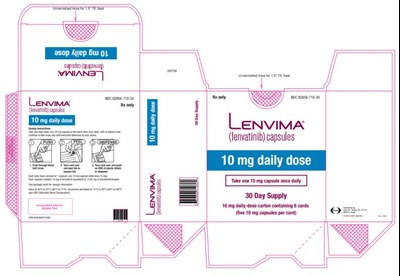
- PRINCIPAL DISPLAY PANELNDC 62856-710-30Lenvima(lenvatinib) capsules10 mg daily dose30 day supply10 mg daily- dose carton containing 6 cards - lenvima 08
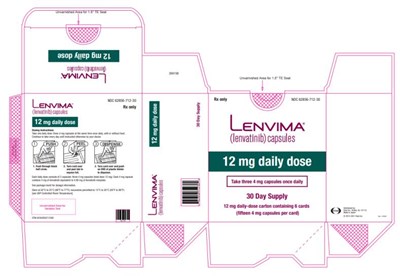
- PRINCIPAL DISPLAY PANELNDC 62856-712-30Lenvima(lenvatinib) capsules12 mg daily dose - lenvima 09
- PRINCIPAL DISPLAY PANELNDC 62856-714-30Lenvima(lenvatinib) capsules14 mg daily dose30 day supply14 mg daily- dose carton containing 6 cards - lenvima 0a
- PRINCIPAL DISPLAY PANELNDC 62856-718-30Lenvima(lenvatinib) capsules18 mg daily dose - lenvima 0b
- PRINCIPAL DISPLAY PANELNDC 62856-720-30Lenvima(lenvatinib) capsules20 mg daily dose30 day supply20 mg daily- dose carton containing 6 cards - lenvima 0c
- PRINCIPAL DISPLAY PANELNDC 62856-724-30Lenvima(lenvatinib) capsules24 mg daily dose30 day supply24 mg daily- dose carton containing 6 cards - lenvima 0d
- NDC 62856-704-05Lenvima(lenvatinib) capsules4 mg daily dose - lenvima 0e
- NDC 62856-708-05Lenvima(lenvatinib) capsules8 mg daily dose - lenvima 0f
- NDC 62856-710-05Lenvima(lenvatinib) capsules10 mg daily dose - lenvima 10
- NDC 62856-712-05Lenvima(lenvatinib) capsules12 mg daily dose - lenvima 11
- NDC 62856-714-05Lenvima(lenvatinib) capsules14 mg daily dose - lenvima 12
- NDC 62856-718-05Lenvima(lenvatinib) capsules18 mg daily dose - lenvima 13
- NDC 62856-720-05Lenvima(lenvatinib) capsules20 mg daily dose - lenvima 14
- NDC 62856-724-05Lenvima(lenvatinib) capsules24 mg daily dose - lenvima 15
- lenvima 16
- lenvima 17
- lenvima 18
- lenvima 19
Product Label Images
The following 25 images provide visual information about the product associated with Lenvima NDC 62856-704 by Eisai Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
The chemical structure of lenvatinib mesylate is LENVIMA, a kinase inhibitor, is the mesylate salt of lenvatinib. Its chemical name is 4-[3-chloro-4-(N’-cyclopropylureido)phenoxy]-7-methoxyquinoline-6 carboxamide methanesulfonate. The molecular formula is C21H19ClN4O4 • CH4O3S, and the molecular weight of the mesylate salt is 522.96. - lenvima 01
![lenvima-01.jpg The chemical structure of lenvatinib mesylate is LENVIMA, a kinase inhibitor, is the mesylate salt of lenvatinib. Its chemical name is 4-[3-chloro-4-(N’-cyclopropylureido)phenoxy]-7-methoxyquinoline-6 carboxamide methanesulfonate. The molecular formula is C21H19ClN4O4 • CH4O3S, and the molecular weight of the mesylate salt is 522.96. - lenvima 01](https://ndclist.com/assets/spl/images/f4bedd21-efde-44c6-9d9c-b48b78d7ed1e/400x-lenvima-01.jpg)
Figure 4: Kaplan-Meier Curves for Overall Survival in REFLECT - lenvima 05

This is a list of medications and dosages. It includes Everolimus at 10mg and a combination of Lenvima (18mg) and Everolimus (Smg), along with other unclear information about subjects and subgroups. There is not enough information to provide further context.*
PRINCIPAL DISPLAY PANELNDC 62856-710-30Lenvima(lenvatinib) capsules10 mg daily dose30 day supply10 mg daily- dose carton containing 6 cards - lenvima 08
PRINCIPAL DISPLAY PANELNDC 62856-714-30Lenvima(lenvatinib) capsules14 mg daily dose30 day supply14 mg daily- dose carton containing 6 cards - lenvima 0a
This appears to be a description of a medication called Lenvima, containing 4mg capsules. The package shown contains a 30 day supply and a daily dose is recommended. However, there is no additional information available due to the incomplete and unreadable nature of the text.*
PRINCIPAL DISPLAY PANELNDC 62856-720-30Lenvima(lenvatinib) capsules20 mg daily dose30 day supply20 mg daily- dose carton containing 6 cards - lenvima 0c
PRINCIPAL DISPLAY PANELNDC 62856-724-30Lenvima(lenvatinib) capsules24 mg daily dose30 day supply24 mg daily- dose carton containing 6 cards - lenvima 0d
NDC 62856-708-05Lenvima(lenvatinib) capsules8 mg daily dose - lenvima 0f
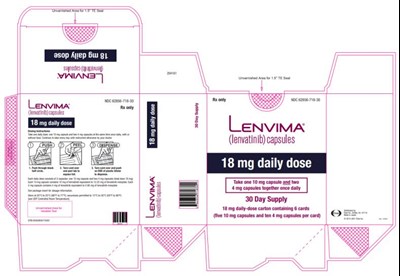
A medicine called "Lenvatinib" is available in capsule form with a daily intake of 18 mg, which contains a total of 68 capsules (8 cards with 10 capsules and 1 card with 8 capsules). It is recommended to take one 10mg capsule and one 4mg capsule daily. The supplied quantity is designed for a 30-day treatment cycle.*
NDC 62856-710-05Lenvima(lenvatinib) capsules10 mg daily dose - lenvima 10

This text appears to be a description of Lenvima capsules, which likely contain the active ingredient lenvatinib. The suggested dose is two 10mg capsules per day, and the package appears to contain a 30 day supply. However, some of the text is not readable and there may be additional important information missing.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.