Product Images Tizanidine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Tizanidine NDC 63187-493 by Proficient Rx Lp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
63187-493-30 - image 01

This is a description of a medication package for Tizanidine 4mg tablets. It contains 90 tablets and is packaged by Proficent Rx LP in Thousand Oaks, CA. The tablets are white to off-white, round, flat, and bevel-edged. Each tablet contains 4.576 mg of tizanidine hydrochloride USP equivalent to 4 mg tizanidine base. The tablets are not interchangeable with Tizanidine capsules. The product ID is PT049390R, and it is manufactured in India by UNICHEM LABORATORIES LTD. The lot number is 00000 and the expiration date is 00/00/00100. The medication should be stored between 20-25°C (68°-77°F) and kept out of the reach of children.*
Figure 2 - tizanidinetabsusp figure 03
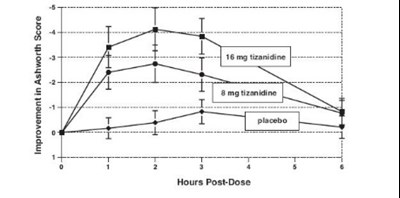
The text seems to describe a comparison between two treatments regarding an improvement in the Ashworth score, a measure of muscle spasticity commonly used in neurological conditions. One of the treatments is indicated by the drug 8 ma tamnidine, although it is unclear what the other treatment is as it is only referenced as "placebo". The results may be presented in a graph or table showing the changes in Ashworth scores over time, as indicated by the "Hours Post-Dose" label.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.



