Product Images Valsartan And Hydrochlorothiazide
View Photos of Packaging, Labels & Appearance
- Figure 1: Probability of Achieving Systolic Blood Pressure <140 mmHg at Week 8 - valhyctz fig1
- Figure 2: Probability of Achieving Diastolic Blood Pressure <90 mmHg at Week 8 - valhyctz fig2
- Figure 3: Probability of Achieving Systolic Blood Pressure <130 mmHg at Week 8 - valhyctz fig3
- Figure 4: Probability of Achieving Diastolic Blood Pressure <80 mmHg at Week 8 - valhyctz fig4
- 63187-571-30 - valhyctz fig5
- Valsartan Chemical Structure - valhyctz str1
- Hydrochlorothiazide Chemical Structure - valhyctz str2
Product Label Images
The following 7 images provide visual information about the product associated with Valsartan And Hydrochlorothiazide NDC 63187-571 by Proficient Rx Lp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1: Probability of Achieving Systolic Blood Pressure <140 mmHg at Week 8 - valhyctz fig1
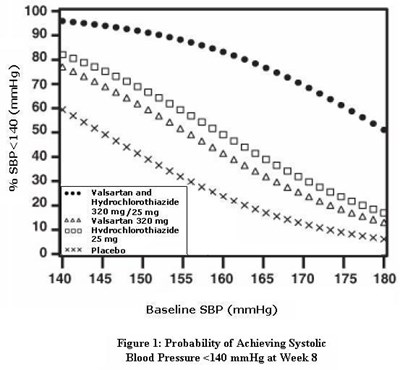
This text appears to be a medical document describing the effectiveness of a medication in lowering systolic blood pressure. The medication is a combination of Valsartan and Hydrochlorothiazide, with a dosage of 320 mg/25 mg. The text presents a graph depicting the probability of achieving a systolic blood pressure level of less than 140 mmHg after 8 weeks of treatment. There are no indications of side effects, contraindications or further instructions.*
Figure 2: Probability of Achieving Diastolic Blood Pressure <90 mmHg at Week 8 - valhyctz fig2
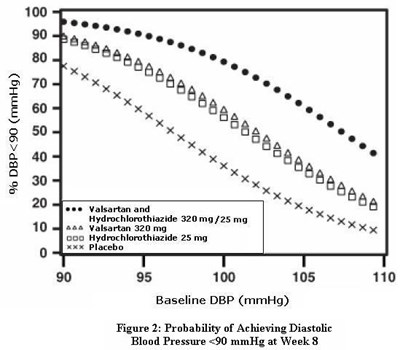
This is a chart representing the probability of achieving a diastolic blood pressure (DBP) below 90 mmHg at week 8. The chart is based on readings of baseline DBP ranging from 95 to 105 mmHg. There is no further information available.*
Figure 3: Probability of Achieving Systolic Blood Pressure <130 mmHg at Week 8 - valhyctz fig3
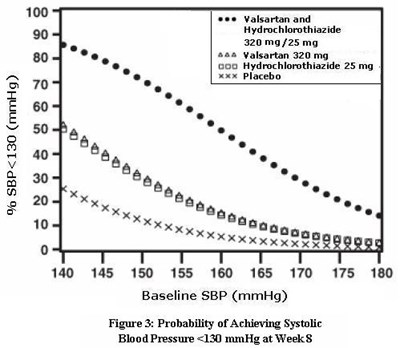
The text describes a figure depicting the probability of achieving systolic blood pressure below 130 mmHg at week 8 for patients taking Valsartan and Hydrochlorothiazide, as well as a placebo. The figure includes a baseline SBP measurement range and dosages for the medications.*
Figure 4: Probability of Achieving Diastolic Blood Pressure <80 mmHg at Week 8 - valhyctz fig4
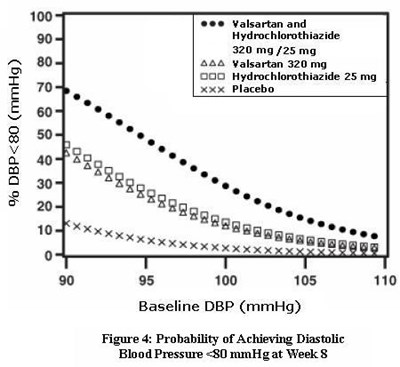
This is a description of a study on the probability of achieving a diastolic blood pressure below 80 mmHg after 8 weeks of treatment with Valsartan and Hydrochlorothiazide at a dosage of 320mg/25mg. The study tested the effectiveness of the medication against a placebo. The graph in Figure 4 shows the probability of achieving the desired blood pressure level.*
63187-571-30 - valhyctz fig5

The text describes a medication called Valsartan HCTZ 80mg/12.5mg. It comes in a bottle of 30 light orange oval tablets. Each tablet contains 80mg of Valsartan USP and 12.5mg of hydrochlorothiazide USP. The tablets are film-coated and have a beveled edge, and are marked with 'I' on one side and '61' on the other. The medication is to be stored between 20-25°C and kept out of reach of children. The lot number is 00000 and the expiration date is 00/00/00. The medication is manufactured by Aurobindo Pharma Limited in India and packaged by Proficient Rx LP in Thousand Oaks, CA.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

