Product Images Duloxetine
View Photos of Packaging, Labels & Appearance
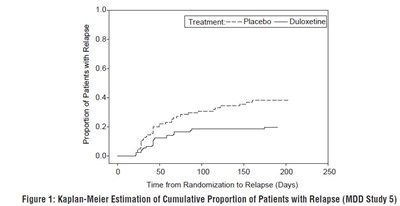
- Figure 1: Kaplan-Meier Estimation of Cumulative Proportion of Patients with Relapse (MDD Study 5) - duloxetine fig1
- 63187-666-30 - duloxetine fig10
- Figure 2: Kaplan-Meier Estimation of Cumulative Proportion of Patients with Relapse (GAD Study 4) - duloxetine fig2
- Figure 3: Percentage of Patients Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity - DPNP-1 - duloxetine fig3
- Figure 4: Percentage of Patients Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity - DPNP-2 - duloxetine fig4
- Figure 7: Percentage of Patients Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity – CLBP-1 - duloxetine fig5
- Figure 8: Percentage of Patients Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity – CLBP-3 - duloxetine fig6
- Figure 9: Percentage of Patients Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity – OA-1 - duloxetine fig7
- Structure - duloxetine str
Product Label Images
The following 9 images provide visual information about the product associated with Duloxetine NDC 63187-666 by Proficient Rx Lp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1: Kaplan-Meier Estimation of Cumulative Proportion of Patients with Relapse (MDD Study 5) - duloxetine fig1
63187-666-30 - duloxetine fig10

This is a medication information sheet for Duloxetine 60mg delayed-release capsules, distributed by Citron Pharma LLC and packaged by Proficient Rx LP. It includes details such as product ID, lot number, expiration date, storage instructions, and the National Drug Code. The medications come in size ‘1’ hard gelatin capsule filled with pellets and are blue/green in colour. The prescription drug is only available by prescription, and each patient should receive a Medication Guide. The medication must be kept out of the reach of children, and it should be stored at 20°-25°C (68°-77°F). The text indicates that the medication is manufactured in India.*
Figure 2: Kaplan-Meier Estimation of Cumulative Proportion of Patients with Relapse (GAD Study 4) - duloxetine fig2

Figure 3: Percentage of Patients Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity - DPNP-1 - duloxetine fig3
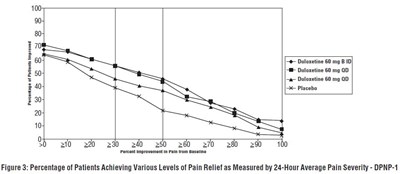
This text describes a clinical trial evaluating the effectiveness of Duloxetine in treating Diabetic Peripheral Neuropathic Pain (DPNP-1). The study compared different doses of Duloxetine with a placebo and measured the percentage of patients experiencing pain relief. The figure shows the percentage of patients achieving various levels of pain relief.*
Figure 4: Percentage of Patients Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity - DPNP-2 - duloxetine fig4

The text describes a graph labeled Figure 4 that displays the percentage of patients that reach various levels of pain relief using Duloxetine 60 mg for diabetic peripheral neuropathic pain. The graph shows results in a 0 to 100% scale and indicates the percentage of improvement from baseline. The graph illustrates the efficacy of the Duloxetine drug with varying levels of pain improvement. No additional useful information was provided.*
Figure 7: Percentage of Patients Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity – CLBP-1 - duloxetine fig5
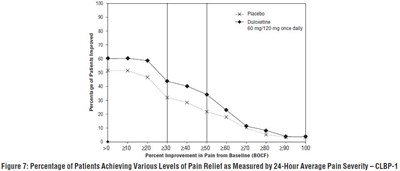
Figure 8: Percentage of Patients Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity – CLBP-3 - duloxetine fig6
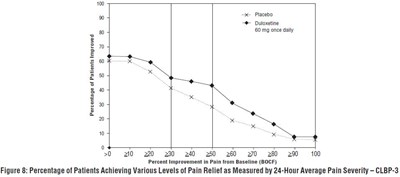
The text describes a figure with a graph displaying percentages of patients achieving various levels of pain relief measured by 24-hour average pain severity. The graph shows different levels of pain improvement from the baseline in percentage. The medication used was Placebo and Duloxetine at a dosage of 60 mg once a day. However, no further details are available to describe the study or the context.*
Figure 9: Percentage of Patients Achieving Various Levels of Pain Relief as Measured by 24-Hour Average Pain Severity – OA-1 - duloxetine fig7
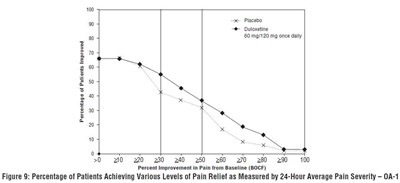
The text appears to be a graph that displays the percentage of patients achieving various levels of pain relief as measured by 24-hour average pain severity. The X-axis shows the percentage improvement in pain from baseline, while the Y-axis shows the percentage of patients. The graph also includes two different treatments, "Placebo" and "Duloxetine," with doses of 0, 60mg/420 mg once daily.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
