Product Images Lenalidomide
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 12 images provide visual information about the product associated with Lenalidomide NDC 63304-046 by Sun Pharmaceutical Industries, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
label 1 - label 1

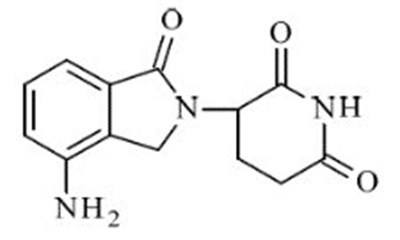
This is a prescription label for Lenalidomide Capsules. The label contains information such as the drug's name, dosage instructions, and NDC number. The label also indicates that the drug may cause birth defects and must be dispensed with an accompanying document.*
spl-lenalidomide-image 08 - spl lenalidomide image 08
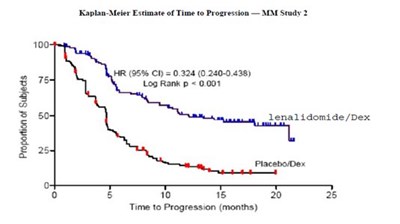
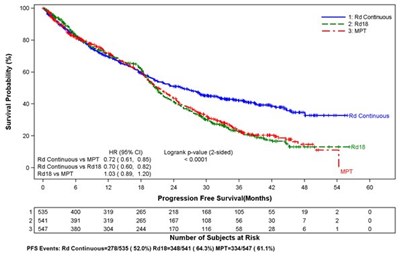
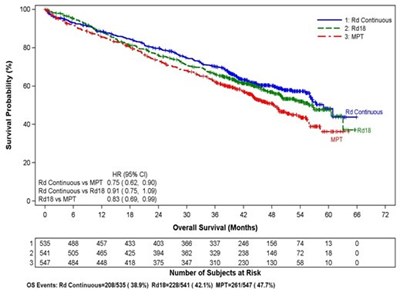
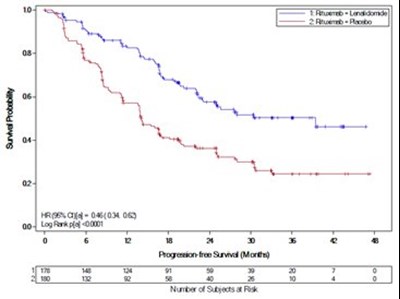
This appears to be a chart or graph displaying the proportion of subjects along with Kaplan-Meler estimates of time to progression for a study involving lenalidomide/Dex versus placebo/Dex. The HR (hazard ratio) for the two treatments is also given.*
spl-lenalidomide-image 5 - spl lenalidomide image 5

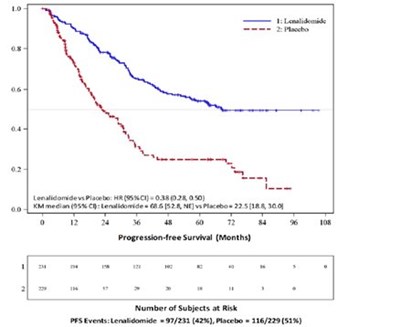
This appears to be a graph or chart showing the proportion of subjects in a study and a Kaplan-Meier estimate of time to progression, specifically for Stage 1. It also indicates a Hazard Ratio and Log Rank test result. The x-axis shows time to progression in months and the y-axis displays the proportion of subjects.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.